Press release
Global GMP Testing Service Market Projected to Grow at a CAGR of 6.5%, Reaching USD 2.49 Billion by 2030, Up from USD 1.5 Billion in 2023
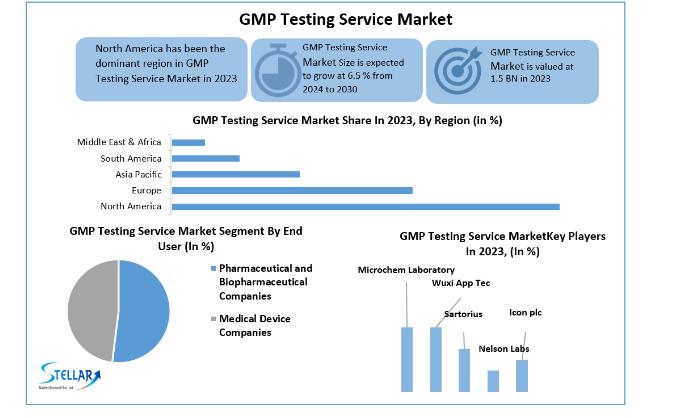
Anticipated Growth in Revenue:The GMP Testing Service Market size was valued at USD 1.5 Bn. in 2023 and the total GMP Testing Service Market revenue is expected to Grow at a CAGR of 6.5% from 2024 to 2030, reaching nearly USD 2.49 Bn. by 2030.
GMP Testing Service Market Overview:
The good manufacturing practice (GMP) system ensures that products are manufactured consistently and under quality standards. The goal is to minimize the risks related to pharmaceutical manufacturing that cannot be eliminated by evaluating the final product. Throughout the projection period, growth is expected in the pharmaceutical and biopharmaceutical firms area. The process of making sure that goods are regularly produced and regulated following quality standards is known as good manufacturing practice, or GMP. It is intended to reduce the dangers associated with producing pharmaceuticals that cannot be removed by testing the finished product.
𝐔𝐧𝐥𝐨𝐜𝐤 𝐈𝐧𝐬𝐢𝐠𝐡𝐭𝐬: 𝐑𝐞𝐪𝐮𝐞𝐬𝐭 𝐚 𝐅𝐫𝐞𝐞 𝐒𝐚𝐦𝐩𝐥𝐞 𝐨𝐟 𝐎𝐮𝐫 𝐋𝐚𝐭𝐞𝐬𝐭 𝐑𝐞𝐩𝐨𝐫𝐭 𝐍𝐨𝐰:https://www.stellarmr.com/report/GMP-Testing-Service-Market/1721
What are GMP Testing Service Market Dynamics?
The pharmaceutical industry allocates a significant portion of its budget to technical developments, market growth in emerging nations, and internal research and development operations. This translates into more money being spent on medication development. The emergence of biosimilars or generic biopharmaceuticals, in addition to new therapies, has led to an increase in the manufacturing of biopharmaceuticals. Generic drug manufacturing has received a lot of attention as a result of the numerous patents that are expiring. To turn promising compounds created in research labs into medications for use by patients, pharma development entails developing, constructing, and scaling up a process.
𝐍𝐞𝐞𝐝 𝐌𝐨𝐫𝐞 𝐈𝐧𝐟𝐨𝐫𝐦𝐚𝐭𝐢𝐨𝐧? 𝐈𝐧𝐪𝐮𝐢𝐫𝐞 𝐀𝐛𝐨𝐮𝐭 𝐒𝐚𝐦𝐩𝐥𝐞 + 𝐆𝐫𝐚𝐩𝐡𝐬 𝐇𝐞𝐫𝐞:https://www.stellarmr.com/report/req_sample/GMP-Testing-Service-Market/1721
What is GMP Testing Service Market Regional Insight?
North America's presence of major players in the market and access to cutting-edge technology is expected to contribute to its considerable market share throughout the forecast period. Additionally, strong medical demand and a supportive regulatory environment are expected to support regional market growth. The FDA constantly monitors medication manufacturers' adherence to its current good manufacturing practice (cGMP) requirements to ensure the quality of their products. Minimum standards for the facilities, procedures, and controls utilized in the production, processing, and packaging of pharmaceutical products are included in the cGMP rules. The rules guarantee that a product is secure and has the components and potency it specifies. Additionally, it is expected that the rising prevalence of chronic illnesses is boost the need for innovative medications, probably resulting in a rise in drug production and market increase.
What is GMP Testing Service Market Segmentation?
By service type
bioanalytical services
Packaging and shelf-life testing
Product validation testing
By end user
pharmaceutical and biopharmaceutical companies
medical device companies
𝐅𝐑𝐄𝐄 𝐆𝐞𝐭 𝐚 𝐂𝐨𝐩𝐲 𝐨𝐟 𝐒𝐚𝐦𝐩𝐥𝐞 𝐑𝐞𝐩𝐨𝐫𝐭 𝐍𝐨𝐰! https://www.stellarmr.com/report/req_sample/GMP-Testing-Service-Market/1721
Some of the current players in the GMP Testing Service Market are:
microchem laboratory
icon plc
sartorius
Wuxi app tec
Nelson labs
Eurofins scientific
Merck kgaa
Ul solution
Intertek group plc
Charles river laboratory
Almac group
namsa.
Key Offerings:
Past Market Size and Competitive Landscape
GMP Testing Service Market Size, Share, Size & Forecast by different segment
Market Dynamics - Growth Drivers, Restraints, Opportunities, and Key Trends by region
GMP Testing Service Market Segmentation - A detailed analysis by Product
Competitive Landscape - Profiles of selected key players by region from a strategic perspective
Competitive landscape - Market Leaders, Market Followers, Regional player
Competitive benchmarking of key players by region
PESTLE Analysis
PORTER's analysis
Value chain and supply chain analysis
Legal Aspects of business by region
Lucrative business opportunities with SWOT analysis
Recommendations
Explore our top-performing reports on the latest trends:
Potato Chips Market https://www.stellarmr.com/report/Potato-Chips-Market/1031
Eco Fiber Market https://www.stellarmr.com/report/Eco-Fiber-Market/1055
Fox Nuts Market https://www.stellarmr.com/report/Fox-Nuts-Market/1105
Ice Cream Market https://www.stellarmr.com/report/Ice-Cream-Market/1168
Semiconductor Bonding Market https://www.stellarmr.com/report/Semiconductor-Bonding-Market/1193
Green Steel Market https://www.stellarmr.com/report/Green-Steel-Market/2124
Contact Stellar Market Research:
S.no.8, h.no. 4-8 Pl.7/4 Kothrud
Pinnac Memories Fl. No. 3
Kothrud, Pune, Maharashtra, 411029
sales@stellarmr.com
+91 9607365656
About Stellar Market Research
Established in 2018, Stellar Market Research is an India-based consulting and advisory firm focused on helping clients reach their business transformation objectives with advisory services and strategic business. The company's vision is to be an integral part of the client's business as a strategic knowledge partner. Stellar Market Research provides end-to-end solutions that go beyond key research technologies to help executives in any organization achieve their mission-critical goals. The company has clients from all across the globe, 23 % from India and others from Japan, Korea, Germany, United States, etc. The company uses its extensive industry knowledge, strong network, and know-how to provide complete insights that give clients a competitive advantage.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global GMP Testing Service Market Projected to Grow at a CAGR of 6.5%, Reaching USD 2.49 Billion by 2030, Up from USD 1.5 Billion in 2023 here
News-ID: 3622987 • Views: …
More Releases from STELLAR MARKET RESEARCH

Ready to Eat (RTE) Baked Products and Bakery Premixes Market Share, Industry Gro …
Ready to Eat (RTE) Baked Products and Bakery Premixes Market was estimated at USD 154.85 Bn. in 2024 and the total revenue is projected to grow at CAGR of 7% From 2025 to 2032 reaching nearly USD 231.41 Bn. by 2032.
Stellar Market Research published a report "Ready to Eat Baked Products and Bakery Premixes Market and forecast (2025-2032)." The report is a comprehensive analysis of global Ready to Eat Baked…

Micromotor Market Comprehensive Research Study, Competitive Landscape and Foreca …
Micromotor Market was estimated at USD 43.10 Bn in 2024, and its total revenue is projected to grow at a CAGR of 7.1% from 2025 to 2032, reaching nearly USD 69.66 Bn by 2032.
Stellar Market Research published a report "Micromotor Market and forecast (2025-2032)." The report is a comprehensive analysis of global Micromotor Market share, size, growth, trends, and forecast. The report also contains competitor analysis and regional analysis and…

Monkeypox Vaccine and Treatment Market Sales and Revenues of Manufacturers Forec …
Monkeypox Vaccine and Treatment Market size was estimated at USD 103.5 Mn in 2024 and the total Global Monkeypox Vaccine and Treatment revenue is projected to grow at a CAGR of 10.20% from 2025 to 2032, reaching nearly USD 225.12 Mn by 2032.
Stellar Market Research published a report "Monkey pox Vaccine and Treatment Market and forecast (2025-2032)." The report is a comprehensive analysis of global monkey pox Vaccine and Treatment…

K12 Education Market Dynamics, Competitive Landscape, Outlook 2025-2032
K12 Education Market Size was valued at USD 2.65 Tn in 2024 and is projected to grow at a CAGR of 12.6% from 2025 to 2032, reaching nearly USD 6.85 Tn by 2032.
Stellar Market Research has released a report "K12 Education Market and forecast (2025-2032)." The report is a detailed analysis of the global market share, size, growth, trends, and forecast of K12 Education Market. It also includes competitor and…
More Releases for GMP
Creative Peptides Released GMP Synthesis Service
Located in Shirley, New York, the world’s leading peptide supplier Creative Peptides announced the launch of its GMP synthesis (https://www.creative-peptides.com/services/custom-gmp-peptide-synthesis-services.html ) business on August 29, 2018. Now this company is focused on the development and GMP manufacturing of pharmaceutical grade peptides.
As the demand of pharmaceutical market continues to grow, more and more pharmas and research institutions choose the CMO and CRO models to expand their businesses, which is more…
Diapharm implements European GMP guidelines in China
Münster (DE), London (UK), Ningbo (CN), 20 December 2013 – Pharmaceutical service provider Diapharm (diapharm.com) is increasing its business activities in China: Diapharm has now implemented a “European” quality management system for Neptune Pharma Ltd (www.neptunepharma.com) in their Joint Venture Partner’s factory in Ningbo, Zhejiang Province. And it has done so successfully: The veterinary medicinal product Trident 500mg/g Powder for Suspension for Fish Treatment (www.trident-50.com), is manufactured onsite under EU…
ECA Foundation releases free GMP WebApp
The ECA Foundation has been providing advanced training and information services in the pharmaceutical industry and especially with regard to pharmaceutical Quality Assurance and GMP compliance for more than 10 years. Now the organisation took advantage of its extensive experience to develop a further free of charge service – the new GMP WebApp.
This new GMP WebApp runs on all smartphones and tablet PCs (Apple and Android platforms) and allows users…
GMP Friction Products Awarded ISO 9001:2008
Internationally Recognized Certification Measures Consistency in Process, Procedure and Quality Performance in Manufacture of Friction Materials
AKRON, OH (March 23, 2011) -- GMP Friction Products, a world leader manufacturing powdered metal friction products for clutch plates and brake pads, recently received certification for ISO 9001:2008.
“ISO 9001:2008 signifies we have taken the extra measure of documenting the policies and standards to ensure consistent compliance with our manufacturing processes,” said Jerry Lynch,…
GMP MANUAL Volume 2 - Validation Procedures by Maas & Peither AG – GMP Publish …
GMP Publishing is launching its new GMP MANUAL Volume 2 – Validation Procedures.
The compendium on validation procedures was written by Dr. Doris Borchert, Dr. Peter Bosshard, Dr. Ralph Gomez, Dr. Michael Hiob, Dr. Christine Oechslein, Max Lazar, Ulrike Reuter, Michael Schulte, Uwe Schwarzat – all international experts and key opinion leaders. They share their detailed understanding of the various aspects of the validation process in clear and comprehensive style…
blue inspection body celebrates 50 GMP audits
Münster (Germany), 20 November 2009. Two years after founding the company and just 18 months after gaining the accreditation blue inspection body GmbH announced today the successful execution of its 50th GMP audit. Further audit trips to China, India, Israel and various European countries have been scheduled already, meaning that in the first quarter 2010 the 75th audit is targeted to be completed. Blue, as a privately organised inspection body,…