Press release
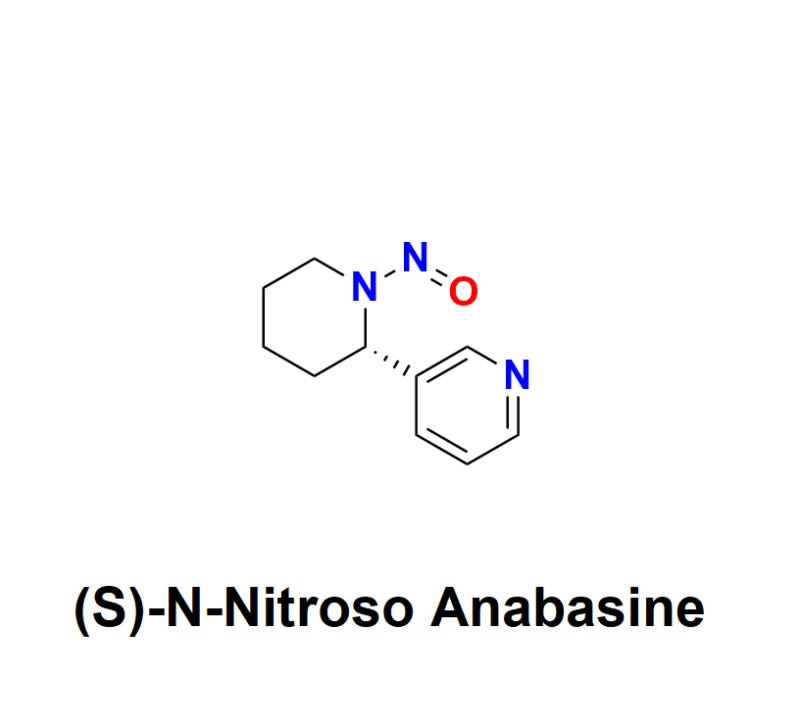
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How Aquigen Bio Sciences Provides Advanced Solutions for Detection, Prevention, and Management to Ensure Pharmaceutical Safety and Compliance
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This reaction can occur during the production, storage, or even formulation of pharmaceuticals. Specifically, (S)-N-Nitroso Anabasine is formed when amines, commonly present in active pharmaceutical ingredients (APIs) or excipients, react with nitrosating agents.
Nitrosamines, including (S)-N-Nitroso Anabasine, are classified as genotoxic impurities, meaning they can interact with DNA and lead to mutations. Over time, this can significantly increase the risk of cancer. Due to these concerns, regulatory bodies like the FDA, EMA, and WHO have demanded more stringent control over nitrosamine impurities in medicines.
Health Hazards of (S)-N-Nitroso Anabasine:
The health risks associated with (S)-N-Nitroso Anabasine are substantial. Studies indicate that chronic exposure, even in minute quantities, can result in:
1. Increased Cancer Risk: Especially cancers of the liver, stomach, and esophagus.
2. Genotoxicity: The ability to damage genetic material, potentially causing long-term cellular dysfunction.
3. Toxic Accumulation: Prolonged ingestion of contaminated medicines may lead to systemic toxicity.
These risks underscore the critical need for pharmaceutical companies to take preemptive measures to identify and eliminate (S)-N-Nitroso Anabasine from their drug products.
Get in Touch with Us - https://aquigenbio.com/contact-us/
The Regulatory Imperative
To combat the dangers posed by nitrosamines, global regulatory bodies have introduced strict guidelines. These include:
1. Risk Assessment: Manufacturers must evaluate all production processes to identify potential sources of nitrosamine contamination.
2. Analytical Detection: Advanced tools like gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-tandem mass spectrometry (LC-MS/MS) are recommended for precise detection.
3. Permissible Limits: Regulators have established daily intake thresholds for nitrosamines to ensure patient safety.
4. Corrective Action: Upon detection of impurities, manufacturers must swiftly implement changes to eliminate or minimize their presence.
Non-compliance with these guidelines can result in severe consequences, including product recalls, regulatory penalties, and reputational harm.
Why Pharmaceutical Companies Must Act:
The presence of (S)-N-Nitroso Anabasine in medicines is not just a regulatory challenge; it is a moral responsibility. Protecting patient health demands a proactive approach, integrating quality control systems and rigorous testing protocols into every stage of the drug development lifecycle.
Pharmaceutical companies must:
1. Invest in Advanced Testing: Deploy cutting-edge analytical techniques to ensure precise detection of (S)-N-Nitroso Anabasine and other nitrosamines.
2. Revamp Manufacturing Processes: Identify and eliminate conditions conducive to nitrosamine formation, such as improper pH levels or inadequate control over raw materials.
3. Enhance Supplier Oversight: Collaborate with suppliers to guarantee that raw materials meet stringent safety standards.
4. Stay Informed: Keep up with evolving regulations and best practices to stay ahead of compliance requirements.
The Broader Implications of Nitrosamine Management:
Addressing the issue of (S)-N-Nitroso Anabasine is more than a compliance exercise; it is a step toward safeguarding public health and maintaining the integrity of pharmaceutical brands. Companies that prioritize safety and transparency in their operations stand to gain long-term trust from patients, healthcare professionals, and regulators.
Conclusion: Aquigen Bio Sciences - Your Partner in Nitrosamine Solutions
In the face of mounting concerns surrounding (S)-N-Nitroso Anabasine, Aquigen Bio Sciences stands out as a trusted partner for pharmaceutical manufacturers. As a leading provider of impurity standards and comprehensive analytical services, Aquigen Bio Sciences offers end-to-end solutions to detect, quantify, and mitigate nitrosamine impurities effectively.
With a team of seasoned experts and cutting-edge technologies, Aquigen Bio Sciences empowers pharmaceutical companies to meet regulatory requirements while safeguarding patient health. From risk assessments to analytical testing and customized solutions, their expertise ensures manufacturers can address the challenges of nitrosamine impurities confidently and effectively.
For more information on how Aquigen Bio Sciences can assist in managing (S)-N-Nitroso Anabasine risks, connect with their team today.
Similar Trending Products:
1) N-Nitroso Sitagliptin Impurity: https://aquigenbio.com/product/n-nitroso-sitagliptin-impurity-2/
2) N-Nitroso N-Desmethyl Nicardipine: https://aquigenbio.com/product/n-nitroso-n-desmethyl-nicardipine/
3) N-Nitroso Apixaban Amino Acid Impurity: https://aquigenbio.com/product/n-nitroso-apixaban-amino-acid-impurity/
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a renowned contract research organization based in Pune, India, specializing in impurity standards and comprehensive solutions for the pharmaceutical industry. With a focus on advancing pharmaceutical safety and compliance, Aquigen provides expert guidance on identifying, quantifying, and managing impurities in various drug formulations. Their commitment to quality and regulatory excellence positions them as a trusted partner for manufacturers striving to meet stringent safety standards and improve patient outcomes.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release (S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How Aquigen Bio Sciences Provides Advanced Solutions for Detection, Prevention, and Management to Ensure Pharmaceutical Safety and Compliance here
News-ID: 3764539 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
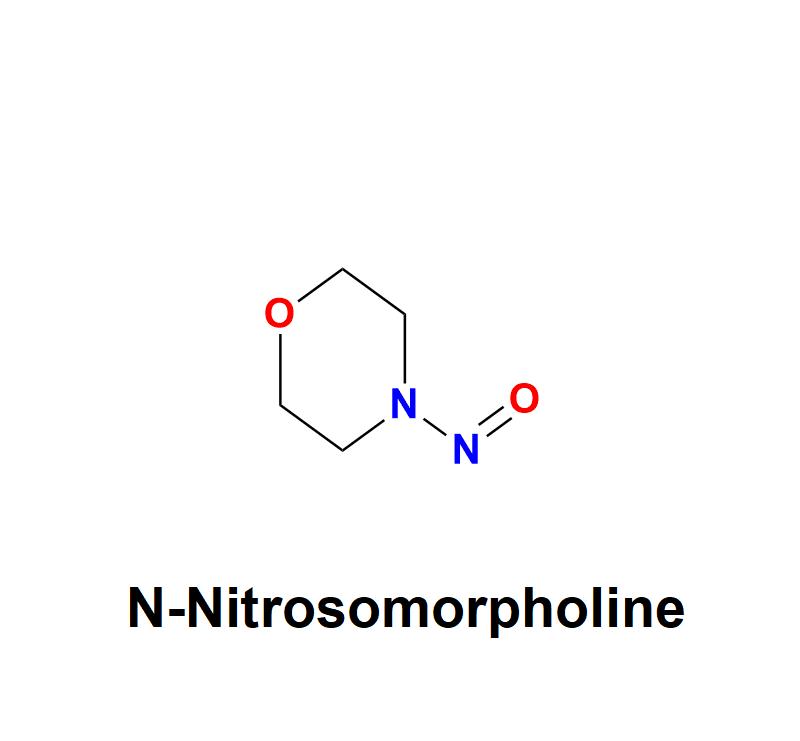
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
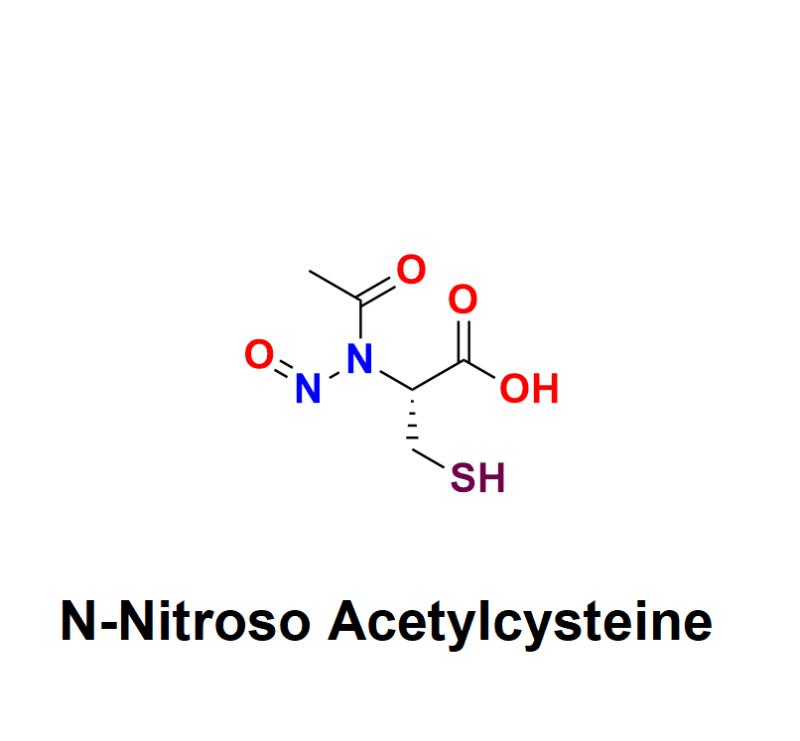
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…
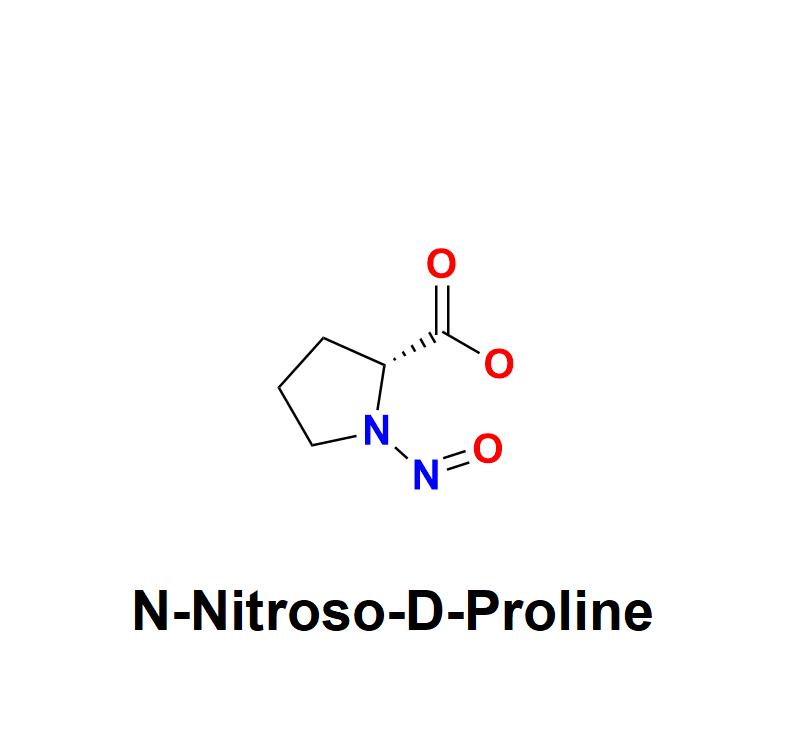
N-Nitroso-D-Proline: A Growing Concern for Pharmaceutical Safety - Explore How A …
N-Nitroso-D-Proline, a chemical impurity belonging to the nitrosamine family, has emerged as a significant concern for pharmaceutical manufacturers worldwide. Its presence in drugs, even at trace levels, poses potential risks to human health due to its genotoxic and carcinogenic properties. As regulatory bodies intensify scrutiny, the pharmaceutical industry must prioritize its detection, mitigation, and prevention to ensure the safety and efficacy of medications.
Learn more about N-Nitroso-D-Proline: https://aquigenbio.com/product/n-nitroso-d-proline/
Understanding N-Nitroso-D-Proline:
N-Nitroso-D-Proline is…
More Releases for Anabasine
Smoking Cessation and Nicotine Addiction Treatment Market 2032: Clinical Trials, …
(Albany, USA) DelveInsight's "Smoking Cessation and Nicotine Addiction - Market Insights, Epidemiology, and Market Forecast - 2034" report delivers an in-depth understanding of the Smoking Cessation and Nicotine Addiction, historical and forecasted epidemiology as well as the Smoking Cessation and Nicotine Addiction market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Smoking Cessation and Nicotine Addiction market report sheds light on…
Smoking Cessation and Nicotine Addiction Market Growth Anticipated by 2034 | Axs …
DelveInsight's "Smoking Cessation and Nicotine Addiction - Market Insights, Epidemiology, and Market Forecast - 2034" report delivers an in-depth understanding of the Smoking Cessation and Nicotine Addiction, historical and forecasted epidemiology as well as the Smoking Cessation and Nicotine Addiction market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
In the market landscape of Smoking Cessation and Nicotine Addiction, an impressive surge…
N-Nitrosodicyclohexylamine Contamination: Challenges for Pharmaceutical Manufact …
The pharmaceutical industry is facing a new safety challenge with the detection of N-Nitrosodicyclohexylamine (NDCHA), a nitrosamine impurity found in various medications. The emergence of NDCHA raises significant concerns for both drug manufacturers and public health, prompting immediate regulatory scrutiny and safety measures.
Understanding N-Nitrosodicyclohexylamine:
N-Nitrosodicyclohexylamine is a nitrosamine compound formed through the reaction of secondary or tertiary amines with nitrite under acidic conditions. Nitrosamines, in general, are classified as probable human…
Anthelmintic Drugs Market Potential Effect on Upcoming Future Growth by Top Vend …
MarketResearchReports.Biz announces addition of new report "Anthelmintic Drugs Market - Global Industry Analysis, Size, Share, Growth, Trends, and Forecast 2018 - 2026" to its database.
Helminths, also known as parasitic worms, are multicellular organisms and can be seen with the naked eye. These parasites are transmitted through soil, bite of vector, and penetration by larva. Soil-transmitted infections are most common and affect nearly 2 billion people in the world, including children.…