Press release
N-Nitroso Impurities: Carcinogenic Risks, Analytical Challenges, and Compliance in Pharmaceuticals
N-Nitroso impurities are emerging as a significant concern in the pharmaceutical industry due to their potential carcinogenicity. These compounds, formed during the manufacturing or storage of drug products, have raised alarms among regulatory agencies across the globe. Pharmaceutical companies face mounting pressure to identify, analyze, and mitigate these impurities through stringent guidelines to ensure patient safety.The serious risks posed by N-Nitroso compounds stem from their ability to induce genetic mutations in living cells, potentially leading to cancer even at trace levels. Regulators like the U.S. FDA, EMA and others have introduced strict limits for the presence of N-Nitroso impurities in active pharmaceutical ingredients (APIs) and drug formulations. As the global focus on drug safety intensifies, pharmaceutical companies are compelled to adopt advanced analytical methods to detect these impurities with unparalleled precision.
To know more about category : https://aquigenbio.com/products/impurity-standards/nitroso/
Carcinogenic Risks of N-Nitroso Impurities
N-Nitroso compounds are classified as probable human carcinogens, which means they carry the potential to cause cancer after prolonged exposure. Their chemical structure, formed through nitrosation reactions of secondary or tertiary amines, makes them inherently reactive. Alarmingly, these impurities can form under a variety of conditions - during synthesis, from excipients, or even during storage if conditions favor nitrosation.
Key examples include N-Nitroso-Clonidine, a representative impurity that has heightened concerns in the pharmaceutical sector due to its structural properties and potential health risks. While significant progress has been made in reducing the presence of these impurities, the challenge remains daunting, as certain pharmaceutical formulations remain particularly susceptible to N-Nitroso formation.
"Mitigating N-Nitroso impurities requires proactive risk assessments, advanced detection methods, and industry-wide collaboration to meet global safety standards and reduce patient risks. This is a priority for the future of pharmaceutical manufacturing", said the CEO of Aquigen Bio Sciences.
To know more about products:
1. N-Nitroso-Clonidine https://aquigenbio.com/product/n-nitroso-clonidine-2/
2.1-Methyl-4-(nitroso)piperazine https://aquigenbio.com/product/1-methyl-4-nitrosopiperazine/
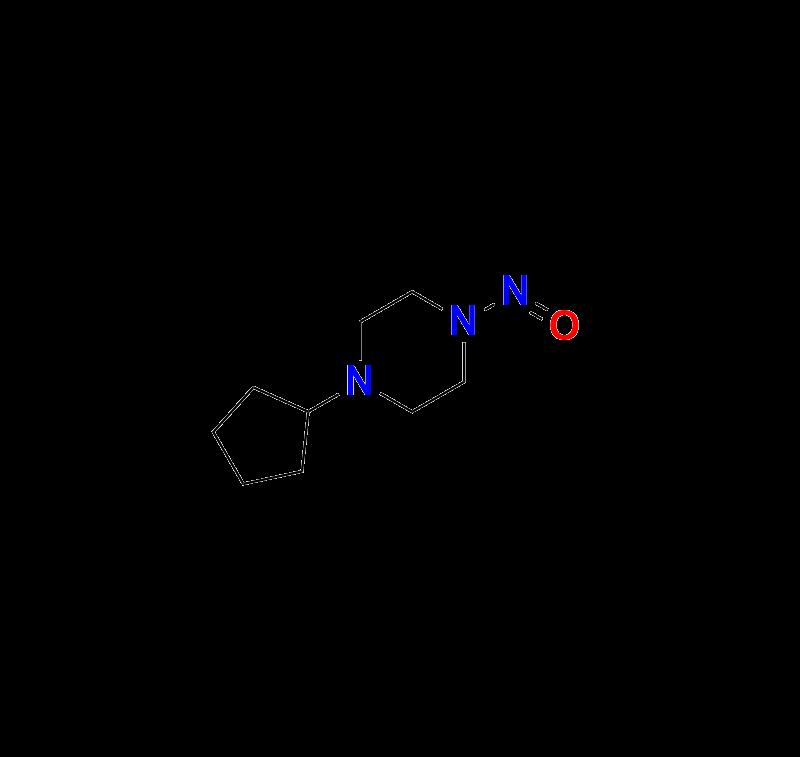
3. 1-Cyclopentyl-4-nitrosopiperazine https://aquigenbio.com/product/1-cyclopentyl-4-nitrosopiperazine/
Analytical Challenges in Detecting N-Nitroso Compounds
The highly volatile and reactive nature of N-Nitroso compounds presents substantial analytical challenges, despite advancements in detection technologies. These compounds are often present in trace amounts, requiring highly sensitive and specific methodologies to detect and quantify them accurately. Analytical techniques such as gas chromatography (GC) paired with mass spectrometry (MS) or liquid chromatography-mass spectrometry (LC-MS) are frequently employed to address this challenge.
For instance, testing for 1-Methyl-4-(nitroso)piperazine, a known impurity in certain drug substances, demands robust instrumentation capable of detecting sub-parts-per-million (ppm) levels. Similarly, the detection of 1-Cyclopentyl-4-nitrosopiperazine, another complex impurity, further emphasizes the need for sophisticated analytical processes. The implementation of these methods not only requires state-of-the-art equipment but a deep understanding of the impurity profiles within specific APIs.
Regulatory Compliance and Industry Action
With heightened public and regulatory scrutiny surrounding N-Nitroso impurities, the pharmaceutical industry has been taking transformative steps to address this issue. Regulatory agencies, including the FDA, EMA, and others, have defined permissible exposure limits for N-Nitroso compounds in pharmaceuticals, mandating strict adherence to these standards.
To tackle this, robust risk assessments are now integrated into the drug development lifecycle, beginning from the sourcing of raw materials through manufacturing processes and storage conditions. This proactive approach is particularly crucial for APIs prone to N-Nitroso formation.
Collaborative Efforts Driving Change
The global effort to combat N-Nitroso impurities extends beyond individual manufacturers. Collaborative initiatives between pharmaceutical companies, regulatory bodies, and research organizations are paving the way for shared knowledge and technological advancement. Industry-wide seminars, workshops, and collaborative projects emphasize the importance of standardizing analytical methods tailored to the unique challenges posed by N-Nitroso compounds.
Standardization has become especially critical given the unpredictability of impurity formation under varied manufacturing conditions. Compliance labs and Contract Research Organizations (CROs) are playing a pivotal role in helping the industry identify and mitigate N-Nitroso contamination, a trend expected to grow as regulations continue to tighten.
Conclusion
The presence of potentially carcinogenic N-Nitroso impurities in pharmaceuticals is a significant challenge that demands immediate and concerted action. By advancing analytical methods, implementing preventive practices, and adhering to stringent regulatory standards, the pharmaceutical industry can safeguard patient safety while maintaining the integrity of drug development processes. Collaborative efforts between manufacturers, regulatory authorities, and scientific organizations remain critical in addressing the complex threat posed by these compounds.
Contact:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences
Aquigen Bio Sciences stands as India's premier resource for high-quality N-Nitroso impurity standards, impelling advancements in pharmaceutical research and manufacturing. Specializing in providing precise impurity standards - including degradation impurities, process impurities, and deuterated isotopes - Aquigen Bio Sciences empowers the industry to comply with the most rigorous global regulations. The contract research organization's commitment to precision and reliability makes it the trusted partner for addressing complex issues like N-Nitroso impurities in pharmaceuticals.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release N-Nitroso Impurities: Carcinogenic Risks, Analytical Challenges, and Compliance in Pharmaceuticals here
News-ID: 3937852 • Views: …
More Releases from Aquigen Bio Sciences

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…

Elevating Analytical Precision with Harmine Impurity Standards
In the realm of pharmaceutical research, controlling the purity of compounds is pivotal. When evaluating psychoactive alkaloids such as harmine-a β-carboline alkaloid known for its reversible monoamine oxidase A inhibition and emerging therapeutic potential-accuracy is non-negotiable. This is where Aquigen Bio Sciences brings its edge: supplying high-grade impurity standards to underpin analytical consistency and regulatory compliance.
Why Harmine Impurity Standards Matter
https://aquigenbio.com/products/impurity-standards/harmine/
Harmine occurs naturally in Peganum harmala and Banisteriopsis caapi, and has…
More Releases for Analytical
Process Analytical Technology Market
Astute Analytica has meticulously crafted the Global Process Analytical Technology Market Research Report with a deep understanding of our client's unique requirements. Our research provides an in-depth analysis of the current regional and global market conditions, delivering valuable insights into the worldwide market landscape. This industry analysis report comprehensively covers market forces, constraints, and their implications for future market demand, offering a broad overview of the sector.
To gain access to…
Analytical Instrumentation Market - Innovative Solutions for Analytical Excellen …
Newark, New Castle, USA: The "Analytical Instrumentation Market" provides a value chain analysis of revenue for the anticipated period from 2023 to 2031. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors.
Analytical Instrumentation Market: https://www.growthplusreports.com/report/analytical-instrumentation-market/8536
This latest report researches the industry structure, sales, revenue,…
Fuel Gas Analyzer Market Latest Trends and Future Growth Study by 2028 | Emerson …
Fuel Gas Analyzer Market: Snapshot
Fuel gas examination is finished utilizing an assortment of gadgets with various elements, for example, some fuel gas analyzers accompany printers that help clients to rapidly and effectively make clear reports on the worksite. The ascending emanation levels and rigid guidelines to control outflow are bringing about the expanding interest for fuel gas analyzers, thus, headways are being made in fuel gas analyzer producers to give…
Pharmaceutical Analytical Testing Outsourcing Market Analysis by 2020-2028 | SGS …
This detailed market report focuses on data from different primary and secondary sources, and is analyzed using various tools. It helps in gaining insights into the market's growth potential, which can help investors identify scope and opportunities. The analysis also provides details of each segment in the global pharmaceutical analytical testing outsourcing market.
Sample Copy of This Report @ https://www.quincemarketinsights.com/request-sample-68060?utm_source=OpenPR&utm_medium=SA
According to the report, the pharmaceutical analytical testing outsourcing market size…
Global Pharmaceutical Analytical Testing Outsourcing Market 2018 Top Key Players …
Summary
WiseGuyReports.com adds “Pharmaceutical Analytical Testing Outsourcing Market 2018 Global Analysis, Growth, Trends and Opportunities Research Report Forecasting to 2023” reports to its database.
This report provides in depth study of “Pharmaceutical Analytical Testing Outsourcing Market” using SWOT analysis i.e. Strength, Weakness, Opportunities and Threat to the organization. The Pharmaceutical Analytical Testing Outsourcing Market report also provides an in-depth survey of key players in the market which is based on the various…
U.S. Analytical Instrument Market Propelled By Tightening Regulatory Pressure On …
The latest report titled “Analytical Instrument Market In The US 2017-2021,” creates a holistic picture of the current situation surrounding analytical tools and equipment and the scope of growth for each key segment over the coming years.
The need to gain a higher level of precision within the various aspects of engineering, especially in terms of consistent monitoring, has given rise to cutting edge developments in the U.S. analytical instrument market.…