Press release
Advancing Precision in Pharmaceutical Development with Resmetirom Impurities: Impurity 13, 13C3 15N, and Impurity 16 from Aquigen Bio
Introduction: Precision Matters in Drug DevelopmentIn today's highly regulated pharmaceutical landscape, the accuracy of analytical research and the integrity of quality control measures have never been more crucial. As pharmaceutical companies continue to develop next-generation therapies, the demand for reliable impurity standards to support method development, validation, and stability studies continues to grow. Among the most promising metabolic drug candidates under research is Resmetirom, a selective thyroid hormone receptor-β (THR-β) agonist currently being evaluated for treating nonalcoholic steatohepatitis (NASH). Given its therapeutic potential, it is imperative for researchers to access high-purity Resmetirom impurity standards for effective analysis and regulatory compliance.
Aquigen Bio, a pioneer in supplying high-quality pharmaceutical impurity standards, is proud to support this critical research with an expanding portfolio of Resmetirom impurities, including Resmetirom Impurity 13, Resmetirom Impurity 13C3 15N, and Resmetirom Impurity 16.
Understanding the Role of Impurity Standards in Resmetirom Research
Impurity standards play a pivotal role in pharmaceutical analysis. During the development and manufacture of active pharmaceutical ingredients (APIs) like Resmetirom, various process-related and degradation impurities can arise. Accurately identifying, quantifying, and characterizing these impurities is a key requirement set forth by global regulatory agencies including the US FDA and the European Medicines Agency (EMA).
Impurities may affect the safety, efficacy, and stability of a drug, which is why validated reference standards like those offered by Aquigen Bio are indispensable for:
Analytical method development and validation
Stability studies
Toxicological assessments
Regulatory filings and DMFs
Quality assurance and control
In the context of Resmetirom, having access to authenticated impurities-especially isotopically labeled standards like 13C3 15N-is essential for robust LC-MS method development and pharmacokinetic studies.
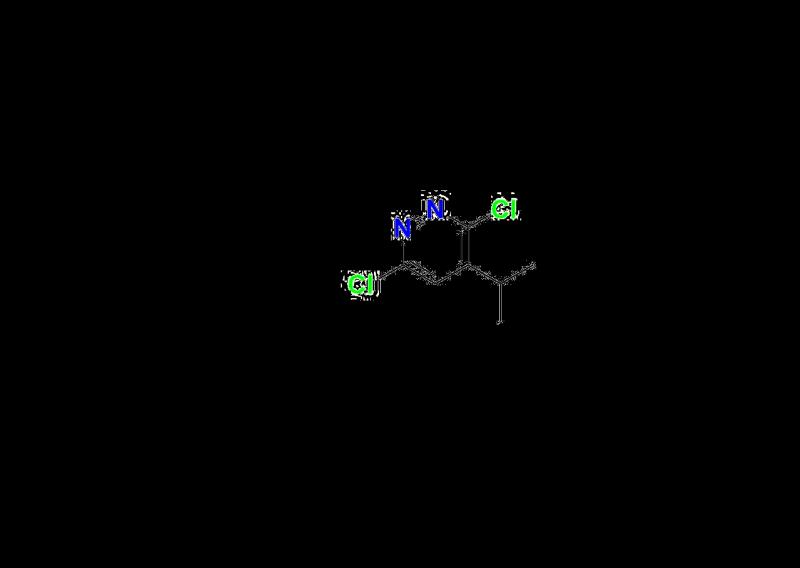
Resmetirom Impurity 13: Supporting Analytical Confidence
View Product - Resmetirom Impurity 13
https://aquigenbio.com/product/resmetirom-impurity-13/
Resmetirom Impurity 13 is one of the key analytical reference materials in Aquigen Bio's impurity portfolio. This impurity helps researchers understand potential synthetic by-products or degradation pathways associated with the production of Resmetirom. Using this standard enables laboratories to:
Accurately quantify Impurity 13 in formulations and bulk materials.
Validate analytical methods for impurity profiling.
Support ICH Q3A/B requirements for impurity identification thresholds.
Aquigen Bio supplies this impurity with detailed characterization data, including HPLC purity, Mass Spectrometry (MS), and Nuclear Magnetic Resonance (NMR) reports, ensuring complete confidence in your analytical outcomes.
Resmetirom 13C3 15N: Isotopically Labeled for Advanced LC-MS Assays
View Product - Resmetirom 13C3 15N
https://aquigenbio.com/product/resmetirom-13c3-15n/
Isotopically labeled standards such as Resmetirom 13C3 15N are essential for high-precision LC-MS/MS quantitation. By incorporating stable isotopes-specifically 13C and 15N-this compound acts as an ideal internal standard during mass spectrometric analysis.
Pharmaceutical and bioanalytical laboratories use this labeled impurity to:
Quantify Resmetirom in biological samples with enhanced sensitivity.
Minimize matrix effects in complex biological fluids.
Support pharmacokinetic, bioequivalence, and toxicokinetic studies.
Improve assay robustness and reproducibility.
Aquigen Bio offers Resmetirom 13C3 15N with certified isotopic enrichment and purity-making it suitable for GLP/GMP compliant environments. Its availability strengthens the analytical infrastructure required for Resmetirom-based drug development.
Resmetirom Impurity 16: Enabling Comprehensive Profiling
View Product - Resmetirom Impurity 16
https://aquigenbio.com/product/resmetirom-impurity-16/
Resmetirom Impurity 16 serves as another critical component in the impurity profile of Resmetirom. Its inclusion in analytical studies helps researchers meet ICH and USP standards for comprehensive impurity profiling and quantification.
Key applications of Resmetirom Impurity 16 include:
Identification and qualification of trace-level degradation products.
Supporting long-term and accelerated stability studies.
Meeting regulatory filing standards for impurity thresholds.
Strengthening control strategies in process development.
Aquigen Bio ensures each batch of Impurity 16 is accompanied by a complete Certificate of Analysis (CoA), including structural confirmation and purity metrics.
Why Choose Aquigen Bio for Resmetirom Impurities?
Aquigen Bio has positioned itself as a trusted global supplier of pharmaceutical reference standards. With a growing catalog of over 2000 impurities and reference materials, the company specializes in hard-to-find, high-purity compounds tailored for advanced pharmaceutical research and development.
Key reasons to choose Aquigen Bio for Resmetirom impurity standards include:
Exceptional Quality: All impurities are synthesized and purified to >95% purity, with accompanying spectral data and CoA.
Regulatory Readiness: Products are designed to support global regulatory submissions with complete documentation.
Quick Turnaround: Most products are available in ready-to-ship quantities, ensuring minimal delays in project timelines.
Custom Synthesis: Aquigen Bio also offers custom synthesis services for unique impurities or isotope-labeled standards not available off-the-shelf.
Global Reach: Serving pharmaceutical manufacturers, CROs, and analytical labs worldwide, Aquigen Bio ensures consistent quality and reliable logistics.
Resmetirom: The Therapeutic Context
Resmetirom (MGL-3196) is gaining attention as a promising THR-β agonist for treating nonalcoholic steatohepatitis (NASH)-a progressive liver disease with limited treatment options. By targeting liver-specific thyroid hormone receptors, Resmetirom aims to reduce liver fat, inflammation, and fibrosis without impacting the cardiovascular system.
As this drug candidate advances through Phase III clinical trials and edges closer to commercial approval, the role of validated impurity standards will only become more critical. Analytical scientists, regulatory teams, and formulation developers must rely on precision tools like Aquigen Bio's Resmetirom impurities to meet the strict demands of safety and efficacy.
The Road Ahead: Supporting Innovation in NASH Treatment
The global pharmaceutical industry is witnessing a surge in research targeting NASH, metabolic disorders, and related liver diseases. As these therapeutic frontiers evolve, so too must the analytical frameworks that support drug development.
Aquigen Bio remains committed to empowering this innovation with:
Expanded impurity libraries for emerging drug candidates like Resmetirom.
Stable isotope-labeled standards for cutting-edge LC-MS technologies.
Expert chemical synthesis capabilities to meet the growing complexity of regulatory and analytical needs.
Explore the Full Range of Resmetirom Impurity Standards
Aquigen Bio offers a comprehensive collection of Resmetirom impurities for pharmaceutical applications. Explore the full Resmetirom category here:
View All Resmetirom Impurities
https://aquigenbio.com/products/impurity-standards/resmetirom/
Whether you're working on impurity profiling, method development, or regulatory filings, Aquigen Bio's specialized impurity standards will support your journey from discovery to compliance.
Contact:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences
Aquigen Bio Sciences is the leading resource for Polyhexanide impurity standards in India. With a commitment to excellence in pharmaceutical research and chemical analysis, Aquigen Bio Sciences provides high-quality impurity standards that support regulatory compliance and quality assurance in pharmaceutical and healthcare industries. Through advanced analytical techniques and rigorous quality control, the company ensures that manufacturers meet the highest safety and efficacy standards in Polyhexanide-based formulations.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Advancing Precision in Pharmaceutical Development with Resmetirom Impurities: Impurity 13, 13C3 15N, and Impurity 16 from Aquigen Bio here
News-ID: 4017685 • Views: …
More Releases from Aquigen Bio Sciences

Elevate Pharmaceutical R&D with Aquigen BioSciences' Precision‐Grade Flibanser …
Flibanserin Impurity B is a reference standard used in pharmaceutical research and development. It is primarily applied during the analysis and validation of drug substances to identify, quantify, and control impurities that may be present in the final product. This impurity is associated with the parent compound, Flibanserin, a medication approved for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women.
Aquigen BioSciences offers Flibanserin Impurity B as a…

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…
More Releases for Resmetirom
Precision in Pharmaceutical Analysis: Resmetirom Impurity Standards from Aquigen …
Resmetirom is a breakthrough thyroid hormone receptor-β agonist, currently under investigation for the treatment of nonalcoholic steatohepatitis (NASH). The successful development, validation, and filing of Resmetirom-based therapies require precision in impurity profiling - a critical cornerstone of global regulatory acceptance.
Why Choose AquigenBio's Resmetirom Impurities?
1. High-Purity Reference Standards: Each impurity is synthesized and purified to >95% purity, with robust documentation including HPLC, MS, and NMR characterization data for your quality assurance.
2.…
Liver Cirrhosis Market Predicted to See Upsurge Through 2034, Highlights DelveIn …
DelveInsight's "Liver Cirrhosis Market Insights, Epidemiology, and Market Forecast-2034 report offers an in-depth understanding of the Liver Cirrhosis, historical and forecasted epidemiology as well as the Liver Cirrhosis market trends in the United States, EU4 (Germany, Spain, Italy, France) the United Kingdom and Japan.
To Know in detail about the Liver Cirrhosis market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Liver Cirrhosis Market Forecast [https://www.delveinsight.com/sample-request/liver-cirrhosis-market?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=gpr]
Some of the key…
Comprehensive Insight into Resmetirom Impurity 11: Analytical Standards, Regulat …
In the intricate world of pharmaceutical manufacturing, ensuring the purity and safety of active pharmaceutical ingredients (APIs) is paramount. Among the various compounds under scrutiny, Resmetirom Impurity 11 has garnered attention due to its significance in the synthesis and quality control processes.
What is Resmetirom Impurity 11?
https://aquigenbio.com/product/resmetirom-impurity-11/
Resmetirom Impurity 11, identified by CAS No. 1581304-48-4, is a chemical by-product that can form during the synthesis of Resmetirom, a therapeutic agent under investigation…
Understanding Resmetirom Impurity 22: A Key to Ensuring Drug Safety and Quality
In the pharmaceutical development process, understanding and controlling impurities is not just a regulatory requirement-it's a cornerstone of patient safety and product efficacy. One impurity garnering increasing attention is Resmetirom Impurity 22, a compound associated with the synthesis of Resmetirom, a promising drug candidate for nonalcoholic steatohepatitis (NASH). This article delves into the significance of Resmetirom Impurity 22, its analytical characterization, implications for pharmaceutical quality control, and how leading suppliers…
Liver Cirrhosis Therapeutics Market Size in the 7MM was ~USD 3,100 Million in 20 …
DelveInsight's "Liver Cirrhosis Market Insights, Epidemiology, and Market Forecast-2034 report offers an in-depth understanding of the Liver Cirrhosis, historical and forecasted epidemiology as well as the Liver Cirrhosis market trends in the United States, EU4 (Germany, Spain, Italy, France) the United Kingdom and Japan.
Discover Key Insights into the Liver Cirrhosis Market with DelveInsight's In-Depth Report @ Liver Cirrhosis Market Size [https://www.delveinsight.com/sample-request/liver-cirrhosis-market?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Key Takeaways from the Liver Cirrhosis Market Report
* The…
Non Alcoholic Fatty Liver Disease (NAFLD) Pipeline Analysis 2024: FDA Approvals, …
(Las Vegas, Nevada, United States) As per DelveInsight's assessment, globally, Non Alcoholic Fatty Liver Disease (NAFLD) pipeline constitutes 80+ key companies continuously working towards developing 100+ Non Alcoholic Fatty Liver Disease (NAFLD) treatment therapies, analysis of Clinical Trials, Therapies, Mechanism of Action, Route of Administration, and Developments analyzes DelveInsight.
"Non Alcoholic Fatty Liver Disease (NAFLD) Pipeline Insight, 2024" report by DelveInsight outlines comprehensive insights into the present clinical development scenario and…