Press release
Ensuring Analytical Excellence and Regulatory Compliance in Oncology Drug Development with High-Purity Pemetrexed EP Impurity D Dihydrate Reference Standard from Aquigen Bi
In pharmaceutical development and manufacturing, the ability to identify, quantify, and control impurities is a cornerstone of drug quality and regulatory compliance. One such critical impurity reference standard is Pemetrexed EP Impurity D Dihydrate, an essential tool for laboratories and manufacturers involved in the production and quality control of Pemetrexed-based drugs.As a leading provider of pharmaceutical impurity standards, Aquigen Bio offers high-purity, well-characterized impurity standards that help ensure your testing processes are accurate, reliable, and aligned with global pharmacopeial requirements.
View the product page for full details and to place an order:
👉 Pemetrexed EP Impurity D Dihydrate - Aquigen Bio
https://aquigenbio.com/product/pemetrexed-ep-impurity-d-dihydrate/
This article provides a deep dive into the significance of Pemetrexed EP Impurity D Dihydrate, its role in pharmaceutical analysis, and its relation to other key impurities like Pemetrexed EP Impurity D, Pemetrexed EP Impurity C, and Pemetrexed EP Impurity A.
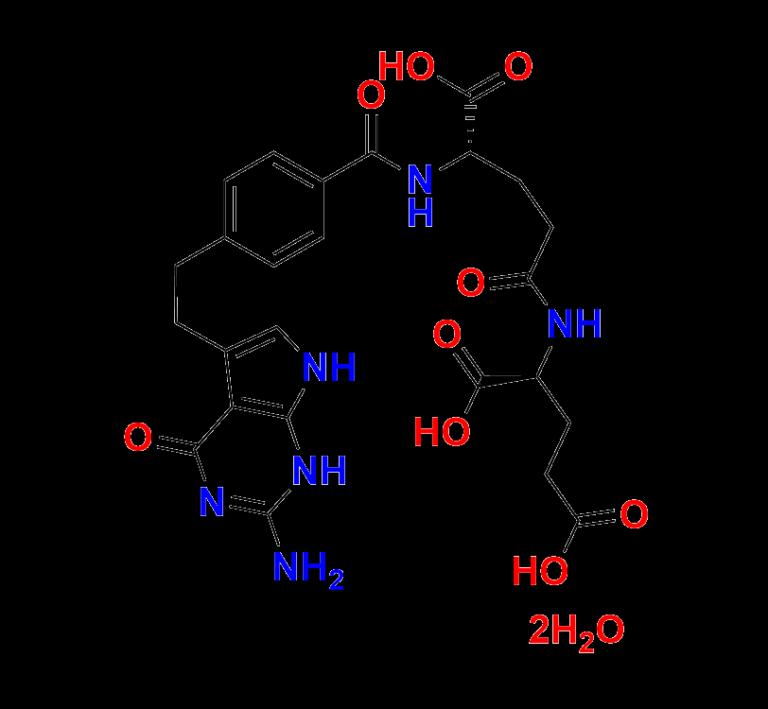
What is Pemetrexed EP Impurity D Dihydrate?
Pemetrexed EP Impurity D Dihydrate is a designated impurity of the chemotherapy agent Pemetrexed, which is primarily used to treat malignant pleural mesothelioma and non-small-cell lung cancer (NSCLC). This dihydrate form of Impurity D is a crystalline solid, representing a hydrated molecular state that is often observed under specific storage or formulation conditions.
The availability of this impurity in its dihydrated state allows analysts and formulators to understand how the active pharmaceutical ingredient (API), Pemetrexed, degrades or transforms over time. More importantly, it plays a vital role in method validation, batch release, and stability testing.
By using Pemetrexed EP Impurity D Dihydrate, pharmaceutical teams can:
Accurately quantify its presence in formulations.
Comply with EP (European Pharmacopoeia) monographs.
Identify degradation patterns during formulation or shelf-life testing.
The Importance of Tracking Impurities in Chemotherapy Drugs
Unlike everyday medications, anticancer drugs like Pemetrexed require extremely stringent testing standards. Small variations in chemical composition or impurity levels can lead to significant changes in drug safety or efficacy.
Impurities are broadly classified into:
Process impurities, arising from the manufacturing process.
Degradation impurities, formed during storage or usage.
Regulatory impurities, outlined in pharmacopeial monographs.
Using certified reference materials like Pemetrexed EP Impurity D Dihydrate ensures that all possible variations are accounted for. With increasing emphasis on ICH Q3A/B guidelines, pharmaceutical manufacturers are required to detect and control even trace-level impurities.
Applications of Pemetrexed EP Impurity D Dihydrate in Pharmaceutical Labs
Analytical Method Development
The presence of dihydrate forms can influence analytical characteristics such as solubility and retention time. Including Impurity D Dihydrate in method development ensures robustness and reproducibility across all testing conditions.
Stability Testing
Dihydrated forms may form under high humidity or temperature variations. Stability studies involving this impurity reveal how environmental conditions affect drug integrity.
Regulatory Submissions
Submitting complete impurity profiles-including hydrated and non-hydrated forms-strengthens the regulatory dossier and accelerates approval timelines.
Batch-to-Batch Consistency
By spiking known quantities of Pemetrexed EP Impurity D Dihydrate, labs can compare batches for consistency and product uniformity.
How Pemetrexed EP Impurity D Dihydrate Differs from Other Impurities
Let's explore the differences and relationships between Pemetrexed EP Impurity D Dihydrate, Impurity D, Impurity C, and Impurity A.
1. Pemetrexed EP Impurity D
https://aquigenbio.com/product/pemetrexed-ep-impurity-d-dihydrate/
Often considered the parent compound of the dihydrate version, Pemetrexed EP Impurity D is a structurally similar degradation product. It exists in an anhydrous form and plays a foundational role in identifying impurity transformation pathways.
Usage: Common in HPLC method development and identification of hydrolysis products.
Relevance: Important for long-term storage analysis and accelerated stability tests.
Aquigen Bio also offers Pemetrexed EP Impurity D for analytical and regulatory purposes, ensuring you have access to the complete impurity profile for your formulations.
2. Pemetrexed EP Impurity C
https://aquigenbio.com/product/pemetrexed-ep-impurity-c/
Pemetrexed EP Impurity C typically arises during dimerization or intermediate-stage synthesis. It is structurally more complex and often studied in conjunction with forced degradation experiments.
Chemical Behavior: May present challenges in detection due to overlapping peaks or low UV absorbance.
Importance: Indicates stress-induced degradation, particularly under thermal or oxidative conditions.
Detecting this impurity helps pharmaceutical companies understand how Pemetrexed behaves under non-ideal storage conditions and informs packaging and formulation decisions.
3. Pemetrexed EP Impurity A
https://aquigenbio.com/product/pemetrexed-ep-impurity-a/
This impurity represents a methylated variant of Pemetrexed. It is most often observed due to methylation in early synthetic stages or through exposure to alkylating agents.
Detection: Sensitive to both UV and MS-based detection systems.
Regulatory Note: Often listed as a reportable impurity in international guidelines.
Tracking Pemetrexed EP Impurity A allows manufacturers to fine-tune their synthetic processes and avoid contamination at the raw material level.
The Aquigen Bio Advantage
Aquigen Bio has built its reputation by supplying highly pure, pharmacopeia-compliant impurity standards to pharmaceutical and biotech companies worldwide. When sourcing Pemetrexed EP Impurity D Dihydrate, clients benefit from:
High Purity Levels (≥98%)
Certificate of Analysis (CoA) for each batch
Same-day dispatch for in-stock items
Technical support for method development and validation
Global Regulatory Relevance
All major regulatory bodies including the US FDA, EMA, and CDSCO encourage the thorough profiling of drug substances and drug products. Having well-characterized impurity standards such as Pemetrexed EP Impurity D Dihydrate aids in:
Satisfying ICH Q3A/B and Q6A guidelines
Meeting specifications outlined in the EP monograph for Pemetrexed
Ensuring compliance in ANDA/NDA submissions
Future Outlook for Impurity Testing in Oncology Drugs
As the pharmaceutical industry moves toward personalized medicine and next-generation oncology treatments, the demand for comprehensive impurity standards will only increase.
Advanced analytical tools like LC-MS/MS, Q-TOF, and NMR require matching standards to confirm structural identities. Impurities like Pemetrexed EP Impurity D Dihydrate will continue to play a central role in ensuring drug quality, patient safety, and regulatory adherence.
Contact:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Us
Aquigen Bio is a leading supplier of high-purity pharmaceutical impurity standards, supporting drug manufacturers and research labs worldwide. We provide certified reference materials for method validation, stability studies, and regulatory compliance-delivering precision, purity, and performance in every product.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Ensuring Analytical Excellence and Regulatory Compliance in Oncology Drug Development with High-Purity Pemetrexed EP Impurity D Dihydrate Reference Standard from Aquigen Bi here
News-ID: 4022049 • Views: …
More Releases from Aquigen Bio Sciences

Elevate Pharmaceutical R&D with Aquigen BioSciences' Precision‐Grade Flibanser …
Flibanserin Impurity B is a reference standard used in pharmaceutical research and development. It is primarily applied during the analysis and validation of drug substances to identify, quantify, and control impurities that may be present in the final product. This impurity is associated with the parent compound, Flibanserin, a medication approved for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women.
Aquigen BioSciences offers Flibanserin Impurity B as a…

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…
More Releases for Pemetrexed
Pemetrexed Injection Market Key Players, Share and Forecast Outlook
"The Pemetrexed Injection Market Is Set To Grow At An Estimated CAGR Of 7.5% From 2025 To 2034, Rising From $1.2 Billion In 2024 To $2.5 Billion By 2034."
On May 15, 2025, Exactitude Consultancy., Ltd. released a research report titled "Pemetrexed Injection Market". This report covers the global Pemetrexed Injection market sales, sales volume, price, market share, ranking of major companies, etc., and provides a detailed analysis by region, country,…
Pemetrexed Market Share, Growth, And Forecast to 2033 Cadila, Abbott, Eli Lilly
Pemetrexed is a chemotherapy drug primarily used to treat non-small cell lung cancer (NSCLC) and malignant pleural mesothelioma. It functions as an antifolate, inhibiting enzymes involved in the synthesis of purines and pyrimidines, which are necessary for DNA and RNA production. This disruption prevents cancer cell proliferation. Pemetrexed is typically given in combination with other chemotherapy agents, like cisplatin, and is administered through an intravenous infusion. It can cause side…
Pemetrexed Market Development Trends & Competitive Analysis by Leading Industry …
Allied Market Research added new research on Pemetrexed Market: Global Opportunity Analysis and Industry Forecast, 2022-2029.
The Pemetrexed Market explores comprehensive study on various segments like size, share, development, innovation, sales and overall growth of major players. The research is based on primary and secondary data sources and it consists both qualitative and quantitative detailing.
Request The Free Sample PDF Of This Report:
https://www.alliedmarketresearch.com/request-sample/10728
Which market perspectives are enlightened in the Pemetrexed Market report?
Executive…
Huateng Pharma Supplies Intermeidates of Pemetrexed Against NSCLC
Pemetrexed disodium is a drug successfully developed by Eli Lilly in the United States for the treatment of tumors, patent protection to January 2017. Pemetrexed disodium is a multi-targeted anti-folate agent with a core pyrrolopyrimidine moiety in its structure. It is a dual inhibitor of nucleotide synthase/dihydrofolate reductase, which inhibits cell replication by disrupting the normal process of folate-dependent metabolism in cells and simultaneously blocking three different enzyme targets critical…
Pemetrexed Disodium API Market Size, Share, Development by 2025
LP INFORMATION recently released a research report on the Pemetrexed Disodium API market analysis and elaborate the industry coverage, current market competitive status, and market outlook and forecast by 2025. Moreover, it categorizes the global Pemetrexed Disodium APImarket by key players, product type, applications and regions,etc.
The main objective of this market research is to help the readers understand the structure of Pemetrexed Disodium APImarket, market definition, overview, industry opportunities…
Pemetrexed Market | Eli Lilly and Company, Stada Arzneimittel AG, Eagle Pharmace …
The Pemetrexed market demand is anticipated to flourish during the forecast period 2020-2027. The report offers information related to import and export, along with the current business chain in the market at the global level. This report provides an in-depth overview of the Pemetrexed market. This includes market characteristics, consisting of segmentation, market share, trends and strategies for this market. The Market Size section provides historical forecasts of market growth…