Press release
Advancing Pharmaceutical Precision: Aquigen Bio's Commitment with Atovaquone EP Impurity A
In today's rapidly evolving pharmaceutical landscape, the demand for accurate impurity profiling and advanced reference standards has never been more vital. Regulatory scrutiny, increased focus on drug efficacy, and safety concerns have all brought impurities into the spotlight. Among these, Atovaquone EP Impurity A stands as a crucial component in the analytical assessment of Atovaquone-based drug formulations. Aquigen Bio, a leading innovator and global provider of certified impurity standards, is setting new benchmarks in pharmaceutical quality with its comprehensive offering of Atovaquone-related impurities.Explore Atovaquone EP Impurity A: https://aquigenbio.com/product/atovaquone-ep-impurity-a/
Understanding Atovaquone and the Role of Impurities
Atovaquone is a hydroxynaphthoquinone antimalarial and anti pneumocystis agent used primarily to treat or prevent Pneumocystis jirovecii pneumonia (PCP) and malaria. As with all active pharmaceutical ingredients (APIs), the presence of impurities in Atovaquone can affect its pharmacological efficacy, safety, and regulatory approval.
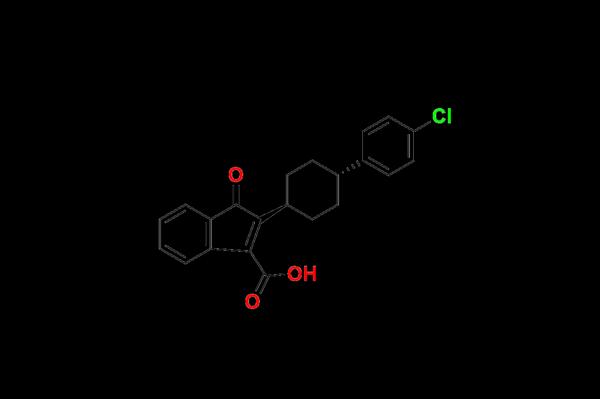
Impurities like Atovaquone EP Impurity A are not mere by-products-they are essential for analytical testing, stability studies, and validation processes during the development and manufacturing of pharmaceuticals. Their accurate characterization ensures drugs are pure, safe, and within permissible limits as per pharmacopeial standards such as those defined in the European Pharmacopoeia (EP).
What is Atovaquone EP Impurity A?
Atovaquone EP Impurity A is a structurally related compound formed during the synthesis or degradation of Atovaquone. The European Pharmacopoeia lists it as one of the critical specified impurities. As a key reference standard, this impurity enables researchers and quality control laboratories to conduct precise quantitative and qualitative assessments of Atovaquone-based formulations.
Aquigen Bio: Driving Quality and Confidence in Impurity Standards
At the heart of Aquigen Bio's mission is an unyielding commitment to pharmaceutical excellence. With a state-of-the-art R&D infrastructure, GMP-aligned production processes, and stringent quality controls, Aquigen Bio delivers reference materials that meet the highest international standards.
The company's extensive catalog includes a wide range of Atovaquone impurities that support pharmaceutical companies across formulation development, regulatory submission, and ongoing stability testing.
In addition to Atovaquone EP Impurity A, Aquigen Bio also offers:
Atovaquone EP Impurity B - Essential for developing a full impurity profile and crucial in method validation.
https://aquigenbio.com/product/atovaquone-ep-impurity-b/
Atovaquone EP Impurity C - A structurally unique impurity providing deeper insight into Atovaquone's degradation pathway.
https://aquigenbio.com/product/atovaquone-ep-impurity-c/
Atovaquone EP Impurity D - Often used in forced degradation and stress testing studies.
https://aquigenbio.com/product/atovaquone-ep-impurity-d/
By offering these certified impurities, Aquigen Bio empowers pharmaceutical professionals with tools that are accurate, reliable, and globally compliant.
Regulatory Importance of Monitoring Impurities
With increasing pressure from global regulatory authorities like the US FDA, EMA, and ICH, impurity profiling has become a non-negotiable component of drug development and registration.
The presence of impurities such as Atovaquone EP Impurity A must be controlled within acceptable limits. Analytical methods must be validated for specificity, precision, linearity, and accuracy using these standards.
Aquigen Bio's high-purity impurity reference materials come with comprehensive Certificates of Analysis (CoA), ensuring consistency, traceability, and confidence for regulatory filings.
The Technical Superiority of Aquigen Bio's Impurity Standards
Aquigen Bio's Atovaquone impurities are synthesized and characterized using advanced techniques including:
High-performance Liquid Chromatography (HPLC)
Mass Spectrometry (MS)
Nuclear Magnetic Resonance (NMR)
Elemental Analysis
FTIR and UV spectroscopy
These analytical characterizations are performed under strictly controlled environments, ensuring reproducibility and traceability. The result? Products like Atovaquone EP Impurity A that not only meet but often exceed pharmacopeial requirements.
Each batch is supported by exhaustive data and documentation, giving end-users absolute confidence in its integrity and reliability.
Applications of Atovaquone EP Impurity A
Pharmaceutical scientists and formulators rely on Atovaquone EP Impurity A for a variety of applications:
Method Development and Validation - Establishing reliable HPLC methods for detecting and quantifying Atovaquone impurities.
Stability Testing - Understanding degradation patterns over time under various stress conditions.
Regulatory Submissions - Meeting ICH Q3A/B and other regional guidelines on impurity control.
Comparative Studies - Benchmarking impurities in innovator and generic drug formulations.
This makes it an indispensable tool in both R&D and quality assurance workflows.
Custom Synthesis and Bulk Availability
Aquigen Bio not only supplies milligram-level quantities for research but also caters to bulk requirements for commercial drug manufacturing. For clients with specialized needs, the company offers custom synthesis and impurity isolation services tailored to specific project requirements.
Whether it's a gram-scale synthesis of Atovaquone EP Impurity A for routine testing or a bespoke impurity profile development, Aquigen Bio's scientists collaborate closely with clients to deliver precision-driven solutions.
Why Leading Pharma Companies Trust Aquigen Bio
Pharmaceutical giants and niche formulation companies alike trust Aquigen Bio for several compelling reasons:
Global Compliance - All reference standards are aligned with EP, USP, ICH, and other regulatory guidelines.
Unmatched Purity - Products like Atovaquone EP Impurity A are typically ≥98% pure, ensuring analytical accuracy.
End-to-End Documentation - CoAs, spectral data, and method development support included with each shipment
Responsive Support - A client-centric approach with quick turnaround times, custom packaging, and technical assistance.
Fast Shipping - Global logistics infrastructure for timely delivery across the US, Europe, Asia, and other key markets.
A Comprehensive Atovaquone Impurity Portfolio
With the growing focus on fixed-dose combinations and global registration filings, having access to a full suite of impurity standards is essential. Aquigen Bio's Atovaquone impurity portfolio supports this need:
Atovaquone EP Impurity A - Structural consistency and high-purity grade for analytical excellence.
Atovaquone EP Impurity B - Important for understanding intermediate synthesis by-products
Atovaquone EP Impurity C - Crucial in stability-indicating method development.
Atovaquone EP Impurity D - A must-have for forced degradation studies and impurity mapping.
This portfolio enables formulation scientists to design comprehensive impurity profiling strategies that meet international expectations.
Partner with Aquigen Bio for Analytical Confidence
Pharmaceutical development is inherently complex, but impurity profiling doesn't have to be. With Aquigen Bio's trusted standards like Atovaquone EP Impurity A, analytical teams gain a dependable ally in their pursuit of quality and compliance.
Whether you're a formulation scientist validating a new method, a QC analyst performing batch release, or a regulatory affairs professional preparing a submission dossier, Aquigen Bio delivers the impurity reference standards you need-precise, compliant, and ready to ship.
Contact:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio
Aquigen Bio is a specialized provider of certified impurity reference standards, working with pharmaceutical companies worldwide to ensure drug quality, safety, and regulatory compliance. With a growing catalog of over 1,000+ impurities, degradation products, metabolites, and stable isotope standards, Aquigen Bio is redefining excellence in pharmaceutical analytics.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Advancing Pharmaceutical Precision: Aquigen Bio's Commitment with Atovaquone EP Impurity A here
News-ID: 4040643 • Views: …
More Releases from Aquigen Bio Sciences

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…

Elevating Analytical Precision with Harmine Impurity Standards
In the realm of pharmaceutical research, controlling the purity of compounds is pivotal. When evaluating psychoactive alkaloids such as harmine-a β-carboline alkaloid known for its reversible monoamine oxidase A inhibition and emerging therapeutic potential-accuracy is non-negotiable. This is where Aquigen Bio Sciences brings its edge: supplying high-grade impurity standards to underpin analytical consistency and regulatory compliance.
Why Harmine Impurity Standards Matter
https://aquigenbio.com/products/impurity-standards/harmine/
Harmine occurs naturally in Peganum harmala and Banisteriopsis caapi, and has…
More Releases for Atovaquone
Aquigen Bio Expands Impurity Catalogue with the Launch of Atovaquone EP Impurity …
May 05, 2025 - Pune, India - Aquigen Bio, a trusted name in the field of pharmaceutical reference standards and impurities, proudly announces the launch of Atovaquone EP Impurity A, a crucial analytical reference material now available for global supply. With this addition to its expanding portfolio, Aquigen Bio continues to support pharmaceutical researchers and formulation scientists in achieving precision, compliance, and reliability in drug development.
Product Page:
https://aquigenbio.com/product/atovaquone-ep-impurity-a/
What Is Atovaquone EP…
Atovaquone And Proguanil Market Size by Type, Application, and Regional Outlook …
USA, New Jersey- According to Market Research Intellect, the global Atovaquone And Proguanil market in the Internet, Communication and Technology category is projected to witness significant growth from 2025 to 2032. Market dynamics, technological advancements, and evolving consumer demand are expected to drive expansion during this period.
The atovaquone and proguanil market is witnessing robust growth, driven by the increasing prevalence of malaria in tropical and subtropical regions. As malaria remains…
Global Atovaquone Market Research Report 2023
Global Atovaquone Market
Atovaquone is used to prevent or treat a serious lung infection called Pneumocystis pneumonia (PCP). This medication helps to stop infection symptoms such as fever, cough, tiredness, and shortness of breath.
The global Atovaquone market was valued at US$ million in 2022 and is anticipated to reach US$ million by 2029, witnessing a CAGR of % during the forecast period 2023-2029. The influence of COVID-19 and the Netherlands-Ukraine War…
Atovaquone and Proguanil Market (2023-2030) Expectation Surges With Rising Deman …
Atovaquone and Proguanil Market Research Report Forecast 2023-2030 has been prepared by experienced and knowledgeable market analysts and researchers. Each section of the research study is specially prepared to explore key aspects of the Industry. Buyers of the report will have access to accurate Porter's 5 Analysis, PESTLE, SWOT, and other types of analysis on the global Atovaquone and Proguanil market. The report aims to provide a thorough and accurate…
Atovaquone and Proguanil Market to Witness Astonishing Growth by 2028
Atovaquone and Proguanil Market was valued at USD 2202 million in 2021 and is expected to grow at a CAGR of 5% between 2022 and 2028.This Atovaquone and Proguanil Market report provides the way to key organizations for the further expansion and business growth. It also sheds light on newly entering competitors and factors that lessening sales rate. Potential areas are spotted out in this market research report for…
Atovaquone and Proguanil Market Analysis by Trends, Technological Advancement, F …
It covers all the advanced data about the global statistics and status briefly. This in-detailed Atovaquone and Proguanil market analysis report of the market scenario provides information about potent competitors and pricing analysis to aid the new comers to compete and survive in the market. It also covers the holistic summary of the market for the period of 2021 to 2027. This report is the result of the information derived…