Press release
Precision in Impurity Profiling: Aquigen Bio Introduces High-Purity Doxorubicin Dimer Impurity 3 for Enhanced Analytical Accuracy
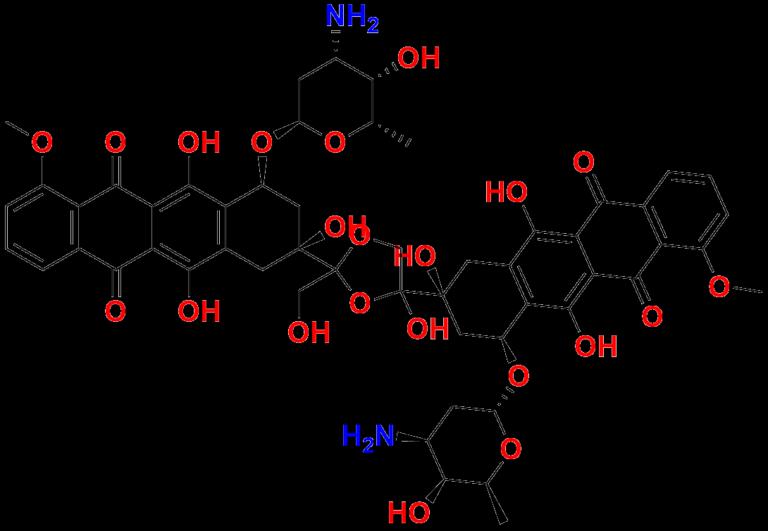
Pune, India - July 2025 - As regulatory standards tighten across the pharmaceutical industry, the need for accurate, high-purity reference standards has never been more critical. Aquigen Bio, a trusted provider of pharmaceutical reference materials, is proud to announce the availability of Doxorubicin Dimer Impurity 3, a key impurity used in the impurity profiling of the widely used chemotherapeutic agent, Doxorubicin.This launch is part of Aquigen Bio's expanded portfolio of Doxorubicin impurity standards, developed to support stringent analytical requirements and regulatory filings for pharmaceutical manufacturers and research organizations around the globe.
What Is Doxorubicin Dimer Impurity 3?
https://aquigenbio.com/product/doxorubicin-dimer-impurity-3/
Doxorubicin Dimer Impurity 3 is a structural byproduct that forms during the synthesis or degradation of Doxorubicin, an anthracycline antibiotic used in the treatment of cancers such as leukemia, breast cancer, and lymphoma. This impurity is generated through oxidative coupling reactions between two Doxorubicin molecules, forming a dimer that may impact the safety, efficacy, and shelf life of the drug product.
With regulatory bodies such as the US FDA, EMA, and CDSCO demanding rigorous impurity profiling, identifying and quantifying dimeric impurities like Doxorubicin Dimer Impurity 3 is essential for regulatory submissions and ensuring product safety.
For analytical chemists and formulation scientists, having access to a well-characterized impurity standard is critical for:
HPLC/UPLC method development and validation
Forced degradation studies
Stability testing
Toxicological assessment
Why Choose Aquigen Bio's Doxorubicin Dimer Impurity 3?
Aquigen Bio delivers this impurity standard with a commitment to scientific excellence and regulatory compliance. Each batch of Doxorubicin Dimer Impurity 3 is synthesized in controlled environments and undergoes stringent quality control and characterization.
Key features include:
≥ 95% purity verified by HPLC and NMR
Detailed Certificate of Analysis (CoA) with spectral data (LC-MS, NMR, IR)
Globally accepted documentation standards
Custom packaging available
Rapid international shipping with cold chain logistics as needed
Aquigen Bio's robust infrastructure, which includes cGMP-compliant synthesis and analytical labs, enables the company to offer consistent, high-quality impurity standards that meet both R&D and regulatory submission needs.
Explore the product in detail: Doxorubicin Dimer Impurity 3
A Full Suite of Doxorubicin Impurity Standards
Aquigen Bio understands that a single impurity standard rarely suffices for a complete analytical profile. To support comprehensive testing and validation, the company offers a broad spectrum of Doxorubicin-related impurity standards, including:
Doxorubicin Dimer Impurity 1 -
https://aquigenbio.com/product/doxorubicin-dimer-impurity-1/
A closely related structural isomer used to assess side reactions and impurity build-up during synthesis or formulation.
Doxorubicin Dimer Impurity 2 -
https://aquigenbio.com/product/doxorubicin-dimer-impurity-2/
Used in stability studies and toxicological risk assessments for advanced Doxorubicin formulations.
These impurities, along with Doxorubicin Dimer Impurity 3, are part of Aquigen's specialized category of Doxorubicin impurity standards that support analytical development, method validation, and regulatory submissions worldwide.
Regulatory Compliance and Documentation
Pharmaceutical companies must ensure that all impurity standards used in their testing protocols meet internationally accepted criteria. Aquigen Bio aligns its manufacturing and quality control practices with ICH Q3A/B, USP, and Ph. Eur. guidelines, delivering reference materials that support:
Drug Master File (DMF) submissions
New Drug Applications (NDAs)
ANDA filings
GMP audits and inspections
Each impurity standard is supplied with a full suite of analytical documentation, including:
COA (Certificate of Analysis)
NMR, MS, IR spectra
HPLC chromatogram
Stability and storage data
This comprehensive documentation simplifies both internal QC processes and external audits or regulatory queries.
Applications Across the Drug Development Pipeline
The availability of high-purity dimer impurities like Doxorubicin Dimer Impurity
Explore Doxorubicin Impurities
https://aquigenbio.com/products/impurity-standards/doxorubicin/
3 facilitates critical pharmaceutical functions:
Method Development: Establishing sensitive, specific methods for impurity detection and quantification.
Stability Studies: Understanding the degradation pathways of Doxorubicin under various environmental conditions.
Quality Control: Monitoring batch-to-batch consistency of active pharmaceutical ingredients (APIs) and finished products.
Toxicological Studies: Assessing the safety of trace-level impurities that could pose potential risks to patients.
Contact:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio
Aquigen Bio is a global leader in pharmaceutical impurity reference standards, offering a comprehensive catalog that spans APIs, degradation products, metabolites, and custom synthesis services. Headquartered in Hyderabad, India, Aquigen Bio serves clients in over 30 countries, including regulatory agencies, pharmaceutical manufacturers, CROs, and academic research institutions.
With a mission to support precision, safety, and scientific innovation, Aquigen Bio continues to invest in cutting-edge analytical capabilities, world-class talent, and stringent quality systems.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Precision in Impurity Profiling: Aquigen Bio Introduces High-Purity Doxorubicin Dimer Impurity 3 for Enhanced Analytical Accuracy here
News-ID: 4093558 • Views: …
More Releases from Aquigen Bio Sciences

Elevate Pharmaceutical R&D with Aquigen BioSciences' Precision‐Grade Flibanser …
Flibanserin Impurity B is a reference standard used in pharmaceutical research and development. It is primarily applied during the analysis and validation of drug substances to identify, quantify, and control impurities that may be present in the final product. This impurity is associated with the parent compound, Flibanserin, a medication approved for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women.
Aquigen BioSciences offers Flibanserin Impurity B as a…

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…
More Releases for Doxorubicin
Rising Prevalence Of Cancer Fuels Growth In Liposomal Doxorubicin Market: A Sign …
The Liposomal Doxorubicin Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Liposomal Doxorubicin Market Size and Projected Growth Rate?
The liposomal doxorubicin market has also shown strong growth in recent years. It is expected to grow from $1.23 billion in 2024 to $1.32…
Doxorubicin Market: Prospects for Long-Term Value & Growth
The "Global Doxorubicin" intelligence report, just published by USD Analytics, covers insurers' micro-level study of important market niches, product offers, and sales channels. In order to determine market size, potential, growth trends, and competitive environment, the Global Doxorubicin provides dynamic views. Both primary and secondary sources of data were used to generate the research, which has both qualitative and quantitative depth. Several of the major figures the study featured Johnson…
Exploring the Doxorubicin Market: A Comprehensive Deep Dive
The Doxorubicin market has been on an impressive growth trajectory, largely propelled by the alarming surge in cancer cases across the globe. As of 2023, the market has already reached a substantial valuation of USD 1,218 million, and experts anticipate a promising future. Projections indicate a Compound Annual Growth Rate (CAGR) of 6.4% during the forecast period spanning from 2024 to 2032. This growth trajectory aims to achieve an astonishing…
Liposomal Doxorubicin Market - Powerful Delivery, Enhanced Outcomes: Empowering …
Newark, New Castle, USA - new report, titled Liposomal Doxorubicin Market The report has been put together using primary and secondary research methodologies, which offer an accurate and precise understanding of the Liposomal Doxorubicin market. Analysts have used a top-down and bottom-up approach to evaluate the segments and provide a fair assessment of their impact on the global Liposomal Doxorubicin market. The report offers an overview of the market, which…
Doxorubicin Market Value Projected to Expand by 2032
From 2022 to 2026, demand for doxorubicin is expected to rise at a 5.3% CAGR. The global doxorubicin market is currently valued at US$ 1.1 billion and is expected to reach US$ 1.3 billion by the end of 2026. Doxorubicin shipments in Japan and Canada are expected to rise at CAGRs of 3.9% and 5.3%, respectively, through 2026.
𝐃𝐨𝐰𝐧𝐥𝐨𝐚𝐝 𝐅𝐫𝐞𝐞 𝐒𝐚𝐦𝐩𝐥𝐞 𝐂𝐨𝐩𝐲 𝐨𝐟 𝐭𝐡𝐢𝐬 𝐑𝐞𝐩𝐨𝐫𝐭: https://www.factmr.com/connectus/sample?flag=S&rep_id=7303
Analysts aim to provide a…
Global Doxorubicin Market Insights, Forecast
Doxorubicin is a kind of antitumor antibiotics, can inhibit the synthesis of RNA and DNA, RNA in the strongest inhibitory effect. Doxorubicin has an effect on a wide variety of tumor, belongs to the cycle non-specific drug, have all sorts of growth cycle of tumor cells to kill.it's given by injection into a vein. The global Doxorubicin market is valued at 910 million US$ in 2018 and will reach 1410…