Press release
Aquigen Bio Expands its Impurity Standards Portfolio with Malathion & Dithiodisuccinate Reference Materials
Aquigen Bio Sciences, a leading pharmaceutical impurity‐standards manufacturer based in India, continues to broaden its catalog with new high‐quality analytical reference standards. These include Malathion EP Impurity A, O,O‐Dimethyl Malathion, and Tetraethyl Dithiodisuccinate, designed to support rigorous research, validation, and regulatory compliance in agrochemical and pharmaceutical analysis.Introducing New Additions to the Impurity Standards Line-up
https://aquigenbio.com/product/tetraethyl-dithiodisuccinate/
Malathion EP Impurity A
This compound-diethyl (2R)-2‐((methoxy(methylthio)phosphoryl)thio)succinate-offers traceability to pharmacopeial standards, such as USP or EP. It is supplied with full characterization data and COA, making it ideal for analytical method development, quality control, and abbreviated new drug applications (ANDA)O,O‐Dimethyl Malathion (also known as O,O‐dimethyl malathion)
Positioned alongside Malathion EP Impurity offerings, O,O‐Dimethyl Malathion serves as a crucial reference impurity standard to help researchers monitor residual pesticide levels during manufacturing and formulation validation
https://aquigenbio.com/product/oo-dimethyl-malathion/
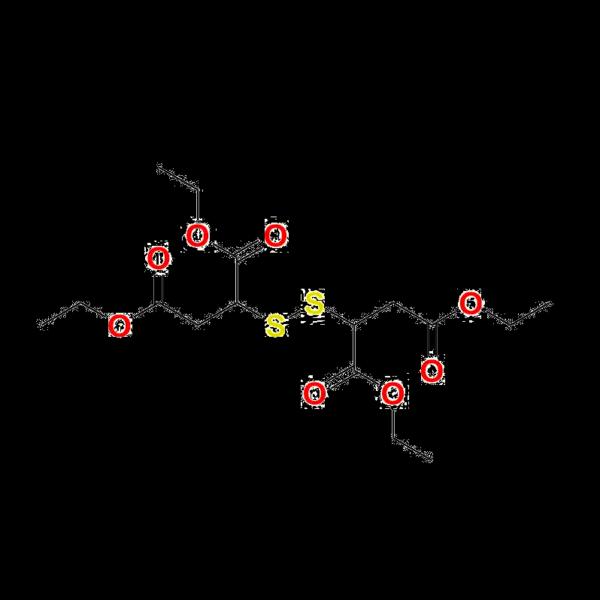
Tetraethyl Dithiodisuccinate
This standard (CAS 2090‐25‐7; molecular formula C16H26O8S2, MW 410.5) is meticulously characterized for accurate analytical use. With COA and traceability features, it supports high-end analytical chemistry workflows and method validation in pharmaceutical or agrochemical R&D
Why Aquigen Bio Standards Set the Benchmark
Comprehensive Documentation: Each product has validated HPLC, MS, and NMR data to ensure reproducibility and regulatory alignment.
Regulatory Compliance Ready: Standards are built for use with pharmacopeial guidelines (USP/EP) and support ANDA, method validation, and QC workflows.
Non‐clinical Use Only: All impurity standards are provided strictly for analytical purposes-not human consumption.
https://aquigenbio.com/product/tetraethyl-dithiodisuccinate/
Centered on Quality and Reliability
Aquigen Bio holds key certifications including ISO‐9001:2015, ISO/IEC 17025:2017, and ISO 17034:2016 accreditation for reference standards manufacturing and analytical testing facilities. Their operation includes custom synthesis, analytical support, impurity isolation & characterization, and specialized services such as nitrosamine quantification and deuterated compounds for PK/PD studies.
In recent months, Aquigen Bio has also strengthened its presence globally through its U.S. facility in New Jersey and expanded API impurities analysis capability-including new high‐purity products for Halcinonide, Ibandronate, and others.
https://aquigenbio.com/product/malathion/
Seamless Access and Support
Researchers and quality control teams can request quotes or samples directly via Aquigen Bio's website. End‐to‐end analytical support-from COA delivery to custom synthesis and regulatory documentation-is available on demand.
By integrating these newly released standards into its catalogue, Aquigen Bio strengthens its commitment to precision, regulatory excellence, and innovation in impurity standards. Whether working in agrochemical pesticide analysis or pharmaceutical impurity control, these products offer the accuracy and documentation needed to meet global expectations.
If you'd like to highlight or promote these new products (Malathion EP Impurity A, O,O‐Dimethyl Malathion, Tetraethyl Dithiodisuccinate) in your own PR or blog, let me know-I can help draft optimized content tailored to your platform and audience.
Aquigen Bio is a leading provider of certified reference standards, impurity profiling compounds, and research chemicals to pharma, biotech, and academic institutions worldwide. With a dedication to excellence and a focus on regulatory readiness, Aquigen empowers scientists to innovate with confidence.
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Visit: www.aquigenbio.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Aquigen Bio Expands its Impurity Standards Portfolio with Malathion & Dithiodisuccinate Reference Materials here
News-ID: 4136206 • Views: …
More Releases from Aquigen Bio Sciences

Elevate Pharmaceutical R&D with Aquigen BioSciences' Precision‐Grade Flibanser …
Flibanserin Impurity B is a reference standard used in pharmaceutical research and development. It is primarily applied during the analysis and validation of drug substances to identify, quantify, and control impurities that may be present in the final product. This impurity is associated with the parent compound, Flibanserin, a medication approved for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women.
Aquigen BioSciences offers Flibanserin Impurity B as a…

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…
More Releases for Malathion
Malathion Production Plant Project Report 2025
Setting up a malathion production plant necessitates a detailed market analysis alongside granular insights into various operational aspects, including unit processes, raw material procurement, utility provisions, infrastructure setup, machinery and technology specifications, workforce planning, logistics, and financial considerations.
IMARC Group's report titled "Malathion Production Plant Cost Analysis Report 2025: Industry Trends, Plant Setup, Machinery, Raw Materials, Investment Opportunities, Cost and Revenue" offers a comprehensive guide for establishing a malathion production plant,…
Malathion Production Process: A Comprehensive Insight
Malathion is an organophosphate insecticide widely utilized in agriculture, public health, and residential pest control. As a critical component in controlling pests, understanding the Malathion production process is essential for businesses, investors, and procurement managers involved in the chemical and agricultural industries. This article provides an extensive overview of the production process, incorporating critical aspects such as cost modeling, pre-feasibility analysis, industrial trends, labor charges, utilities, logistics, and supply chain…
Malathion Manufacturing Plant Report 2023: Industry Trends and Machinery
IMARC Group's report, titled "Malathion Manufacturing Plant Project Report 2023: Industry Trends, Plant Setup, Machinery, Raw Materials, Investment Opportunities, Cost and Revenue," provides a complete roadmap for setting up a malathion manufacturing plant. The report covers various aspects, ranging from a broad market overview to intricate details like unit operations, raw material and utility requirements, infrastructure necessities, machinery requirements, manpower needs, packaging and transportation requirements, and more.
In addition to the…
The Malathion Material market to Cascade the “Success” Trove
Malathion is an organophosphate insecticide and specifically an organophosphate parasympathomimetic, which binds to the enzyme group cholinesterase. Malathion products are usually available in the form of dusts, liquid, powder or emulsions. Their toxicity was believed to only affect pests and insects. In the past few decades however, malathion was found to exhibit low levels of human toxicity as well. When used with caution and in the right amount, mosquito…
Global Malathion Material Market Expected to Witness a Sustainable Growth over 2 …
Market Research Report Store offers a latest published report on Malathion Material Market Analysis and Forecast 2019-2025 delivering key insights and providing a competitive advantage to clients through a detailed report.
This report focuses on the key global Malathion Material players, to define, describe and analyze the value, market share, market competition landscape, SWOT analysis and development plans in next few years.
To analyze the Malathion Material with respect to individual growth…
Global Malathion Material Market Snapshot by 2021
Malathion is an organophosphate insecticide and specifically an organophosphate parasympathomimetic, which binds to the enzyme group cholinesterase. Malathion products are usually available in the form of dusts, liquid, powder or emulsions. Their toxicity was believed to only affect pests and insects. In the past few decades however, malathion was found to exhibit low levels of human toxicity as well. When used with caution and in the right amount, mosquito and…