Press release
Biosimilars And Follow-On Biologics Market Forecast and Segments, 2016-2026
Biosimilars is defined as a type of biological product that is similar to another drug, which has already been licensed (approved) by the US FDA or European Medicines Agency. Due to their high degree of similarity with the biological reference product, they have no clinically evidenced and meaningful differences from the reference product in terms of quality, safety or efficacy. These drugs are also coined as follow-on biologics and are mostly derived from biological sources such as bacterium and yeast. The constitution of the biosimilar drugs can be either small molecules such as human insulin or erythropoietin, or complex molecules such as monoclonal antibodies. Biosimilars are increasing gaining prominence given the loss of exclusivity of big branded drugs. In Europe, biosimilars can be marketed through independent applicant following expiry of patent and market exclusivity periods of the reference product. A good example for this is Pfizer acquisition of Hospira, to gain access to the latter’s attractive biosimilars portfolio. Regulatory harmonization, naming and labelling, innovative licensure norms and route to market for the biosimilar drugs are issues expected to gain attention and traction from big drug makers in the forthcoming years.Biosimilars and Follow-on Biologics Market: Drivers and Restraints
Drivers for the biosimilars market include big brand name drugs losing patent extensions, cuts in healthcare costs across nations, forming of incentivized pricing policies by companies in order to access high growth pharmerging markets and good development in pharmacovigilance procedures across the globe. Other factors increasing the demand for biosimilar drugs include rising disease incidences across the globe and better access to healthcare for all nations.
Request Report Sample@ http://www.futuremarketinsights.com/reports/sample/rep-gb-1250
Restraints for the market include constraints in developing and registering biosimilar drugs and the complexity in the manufacturing processes. The drugs are difficult to verify and have to undergo complex regulations. Further, the drugs are different from each other due to variability in raw material and in the manufacturing process, which is expected to deter the future development of biosimilar drugs. There are also risks in having an increase in the versions of an identical biosimilar drug.
Biosimilars and Follow-on Biologics Market: Segmentation
Biosimilars and Follow-on Biologics Market can be segmented into these following ways:
Segmentation by product class
epoetins;
filgrastims;
insulins;
growth hormones;
alfa interferons;
monoclonal antibodies;
beta interferons;
follitropins;
low-molecular-weight heparins (LMWH).
Segmentation by application
Rheumatoid arthritis
Anemia
Cancer
Diabetes
Others
Segmentation by regions
Request For TOC@ http://www.futuremarketinsights.com/toc/rep-gb-1250
Biosimilars and Follow-on Biologics Market: Overview
Biosimilar market is expected to gain prominence over the forthcoming years due to leading biologic drugs expected to lose exclusivity over the next seven years. Further, biosimilar drugs, once formed, are expected to generate cost savings for the patient population. Competition is expected to be limited in the market as the drugs are expected to be formed using various types of innovative technologies. Biosimilars may generate smaller savings for drug makers because of their complexity as well as regulatory challenges of getting FDA approvals.
Biosimilars and Follow-on Biologics Market: Region-wise Outlook
Depending on geographic regions, biosimilar drug market is segmented into seven key regions: North America, South America, Eastern Europe, Western Europe, Asia Pacific, Japan, and Middle East & Africa.
In terms of geography, Europe dominates the market, followed by Asia-Pacific. However, rising technological advancement in healthcare and systematic drug review process will drive the markets over North America, Japan and other regions. Europe dominates the biosimilars market driven by technically advanced healthcare infrastructure and high patient awareness & regulatory harmonization. Increasing funding for development of biosimilar drugs, availability of high-quality research infrastructure and strategies developed by drug makers to restrict entry of new players. Emerging markets include Eastern European countries followed by countries in Eastern Africa. Rising disease incidences in these countries is expected to prove favorable for the growth of the biosimilar drug market.
Biosimilars and Follow-on Biologics Market: Key Players
Some of the key players in biosimilar market are Pfizer Inc. (AC. Hospira), Sandoz International GmbH, , Teva Pharmaceutical Industries Ltd., Dr. Reddy’s Laboratories, Biocon Limited, Mylan, Inc. , Amgen, Celltrion Inc., Roche Diagnostics, and Merck KGaA.
ABOUT US:
Future Market Insights (FMI) is a leading market intelligence and consulting firm. We deliver syndicated research reports, custom research reports and consulting services, which are personalized in nature. FMI delivers a complete packaged solution, which combines current market intelligence, statistical anecdotes, technology inputs, valuable growth insights, an aerial view of the competitive framework, and future market trends.
CONTACT:
616 Corporate Way, Suite 2-9018,
Valley Cottage, NY 10989,
United States
T: +1-347-918-3531
F: +1-845-579-5705
Email: sales@futuremarketinsights.com
Press: press@futuremarketinsights.com
Website: www.futuremarketinsights.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Biosimilars And Follow-On Biologics Market Forecast and Segments, 2016-2026 here
News-ID: 471837 • Views: …
More Releases from Future Market Insights

Aerospace Fluid Conveyance System Market to Reach USD 4.3 Billion by 2035 with I …
The global Aerospace Fluid Conveyance System Market is witnessing steady growth, driven by the increasing demand for advanced fluid handling solutions across commercial and military aircraft. Valued at USD 3.1 billion in 2025, the market is projected to reach USD 4.3 billion by 2035, registering a healthy CAGR of 3.4%. Both established and emerging manufacturers are actively innovating to meet the evolving needs of the aviation sector.
Market Dynamics and Growth…

Automotive Starter and Alternator Market to Reach USD 45.4 Billion by 2035 Amid …
The global Automotive Starter and Alternator Market is on track for substantial expansion, driven by rapid advancements in vehicle technology and the growing focus on electrification. Valued at USD 29.3 billion in 2025, the market is projected to reach USD 45.4 billion by 2035, registering a CAGR of 4.5% during the forecast period. The growth reflects the automotive industry's continuous evolution toward fuel-efficient and sustainable systems that integrate reliability, performance,…
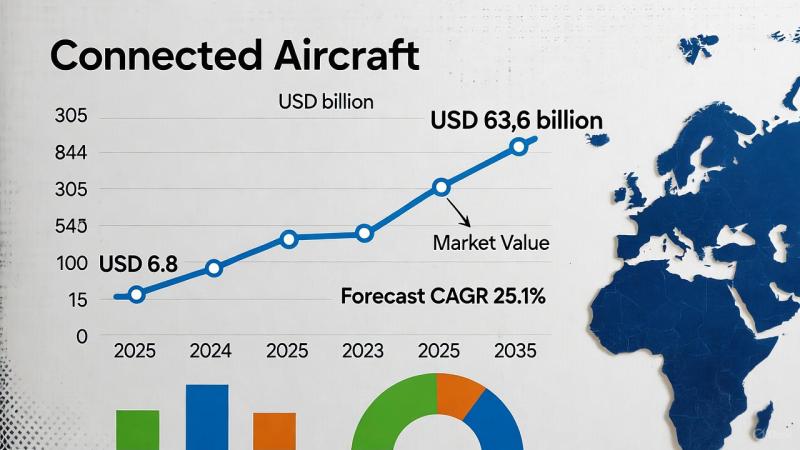
Connected Aircraft Market Soars to USD 63.6 Billion by 2035, Driven by Connectiv …
The global Connected Aircraft Market is poised for transformative growth, projected to rise from USD 6.8 billion in 2025 to USD 63.6 billion by 2035, registering an impressive CAGR of 25.1% over the forecast period. This surge is being fueled by airlines' growing focus on enhancing operational efficiency, passenger experience, and safety through advanced connectivity solutions.
Industry leaders such as Honeywell International Inc., Thales Group, Raytheon Technologies Corporation, BAE Systems PLC,…
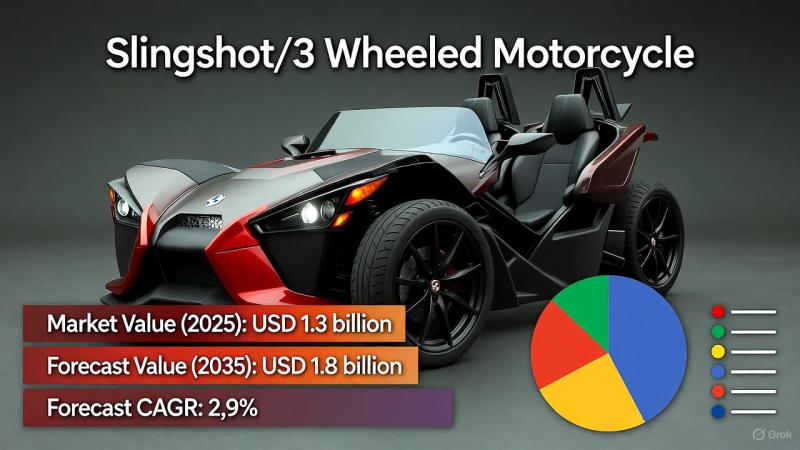
Slingshot/3-Wheeled Motorcycle Market Set to Hit USD 1.8 Billion by 2035 Driven …
The global Slingshot/3-Wheeled Motorcycle Market is on track to witness sustained growth, driven by shifting consumer lifestyles, urban mobility trends, and a rising interest in alternative transportation. Valued at USD 1.3 billion in 2025, the market is projected to reach USD 1.8 billion by 2035, expanding at a CAGR of 2.9%.
During the early growth phase (2021-2025), the market experiences gradual expansion, fueled by consumers' growing appetite for distinctive vehicles that…
More Releases for Biosimilar
Key Trend Reshaping the Biosimilar Monoclonal Antibodies Market in 2025: Advance …
What Are the Projections for the Size and Growth Rate of the Biosimilar Monoclonal Antibodies Market?
In recent times, the biosimilar monoclonal antibodies sector has experienced a swift expansion. The market size, which stands at $8.04 billion in 2024, is projected to climb to $9.25 billion in 2025, marking a compound annual growth rate (CAGR) of 15.1%. Factors such as expired patents, an increased understanding of biosimilars, governmental strategies, heightened financial…
Key Trend Reshaping the Biosimilar Monoclonal Antibodies Market in 2025: Advance …
What Are the Projections for the Size and Growth Rate of the Biosimilar Monoclonal Antibodies Market?
In recent times, the biosimilar monoclonal antibodies sector has experienced a swift expansion. The market size, which stands at $8.04 billion in 2024, is projected to climb to $9.25 billion in 2025, marking a compound annual growth rate (CAGR) of 15.1%. Factors such as expired patents, an increased understanding of biosimilars, governmental strategies, heightened financial…
Biosimilar Market Treating More for Less: The Booming Infliximab Biosimilar Mark …
Infliximab Biosimilar Market worth $ XX Million by 2030 - Exclusive Report by InsightAce Analytic
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Infliximab Biosimilar Market- by Application (Crohn's Disease, Psoriatic Arthritis, Rheumatoid Arthritis, Ulcerative Colitis, Ankylosing Spondylitis, Plaque Psoriasis and Others), End User (Hospital Pharmacy, Retail Pharmacy, Online Pharmacy and Other Direct Distribution Channels), Trends, Industry Competition Analysis, Revenue and Forecast To 2030."
Get…
Biosimilar Monoclonal Antibodies Market
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the " "Global Biosimilar Monoclonal Antibodies Market by Product (infliximab, trastuzumab, rituximab, adalimumab, bevacizumab, cetuximab, ranibizumab, denosumab, eculizumab, and other pipeline products), Indication (oncology, inflammatory & autoimmune disorders, chronic diseases, blood disorders, and other indications), Clinical Trial/Pipeline Analysis, Future Trends, Industry Competition Analysis, Revenue and Forecast To 2031."
The Biosimilar Monoclonal Antibodies Market Size is valued at 5.02…
Infliximab Biosimilar Insight, 2023 | DelveInsight
DelveInsight's, "Infliximab Biosimilar Insight, 2023" report provides comprehensive insights about 35+ companies and 45+ marketed and pipeline drugs in Infliximab Biosimilars landscape. It covers the marketed and pipeline drug profiles, including clinical and nonclinical stage products. It also covers the therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Interested to know more about the functioning of…
Infliximab Biosimilar Insight, 2022 | DelveInsight
DelveInsight's, "Infliximab Biosimilar Insight, 2022" report provides comprehensive insights about 35+ companies and 45+ marketed and pipeline drugs in Infliximab Biosimilars landscape. It covers the marketed and pipeline drug profiles, including clinical and nonclinical stage products. It also covers the therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Interested to know more about the functioning of…
