Press release
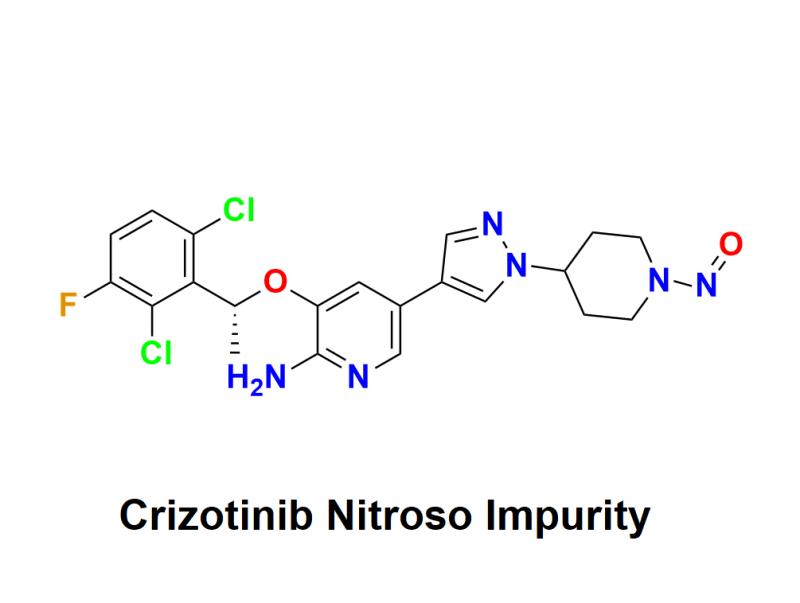
Crizotinib Nitroso Impurity: Understanding the Potential Risks, Safety Concerns, and Their Impact on Cancer Treatment Protocols, Patient Outcomes, and Long-Term Safety Standards in Oncology Care
Crizotinib, a life-saving drug widely used in the treatment of certain types of cancer, particularly non-small cell lung cancer (NSCLC), is at the centre of a recent discovery that has raised significant concerns within the pharmaceutical industry and medical community. The presence of nitroso impurities in Crizotinib has come to light, sparking discussions about the potential risks, impact on cancer treatment protocols, and overall patient safety. This press release aims to explore these emerging concerns and their broader implications for the medical world.Understanding Crizotinib Nitroso Impurity:
Nitroso compounds, such as N-nitrosodimethylamine (NDMA) and N-Nitrosodiethylamine (NDEA), are classified as probable human carcinogens, meaning they have the potential to cause cancer. These impurities can form during drug manufacturing processes or as a result of chemical reactions between active pharmaceutical ingredients (APIs) and excipients. In recent years, nitroso impurities have been detected in several widely used drugs, prompting global recalls and raising alarm over pharmaceutical quality control measures.
Learn more about Crizotinib Nitroso Impurity: https://aquigenbio.com/product/crizotinib-nitroso-impurity/
Crizotinib, a tyrosine kinase inhibitor (TKI), is a targeted therapy designed to block specific proteins that help cancer cells grow. It has proven to be highly effective for patients with ALK-positive NSCLC and other cancers. However, the discovery of nitroso impurities in Crizotinib has raised critical questions about drug safety, manufacturing oversight, and the potential impact on patient outcomes.
Potential Health Risks:
The presence of crizotinib nitroso impurities in medications is particularly concerning because of their carcinogenic nature. Long-term exposure to even low levels of these impurities can increase the risk of developing cancer. For patients already undergoing cancer treatment, the risk becomes more complex. While Crizotinib is intended to treat cancer, the presence of nitroso impurities may inadvertently expose patients to additional cancer-causing agents, leading to significant health risks.
It is essential to note that the level of risk depends on the duration of exposure and the concentration of nitroso impurities in the drug. Regulators such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have set acceptable limits for nitroso impurities in pharmaceuticals. However, any detection of these compounds requires careful evaluation to determine whether the drug remains safe for use.
Impact on Cancer Treatment Protocols:
The discovery of crizotinib nitroso impurities has potential ramifications for cancer treatment protocols worldwide. Crizotinib has been a cornerstone in the treatment of patients with ALK-positive NSCLC, offering them a targeted, effective therapy option. The presence of nitroso impurities, however, may force healthcare providers to reconsider their treatment plans.
Medical practitioners may be required to weigh the benefits of continuing Crizotinib treatment against the potential risks posed by nitroso impurities. In some cases, they may explore alternative therapies, although this could disrupt treatment regimens and affect patient outcomes. Oncologists and healthcare providers need to be fully informed of the situation to make well-considered decisions in the best interest of their patients.
Regulatory Response and Industry Action:
In response to the detection of crizotinib nitroso impurities, regulatory bodies worldwide are conducting thorough investigations to assess the extent of contamination and ensure that drug safety standards are upheld. The FDA, EMA, and other health authorities have launched probes into Crizotinib manufacturing processes to identify the sources of these impurities and implement corrective measures.
Pharmaceutical companies are also taking swift action to address this issue. Manufacturers of Crizotinib are reviewing their production methods to prevent the formation of nitroso impurities and ensure the drug's safety. In some cases, this may involve reformulating the drug or improving quality control measures during manufacturing.
Get in Touch with Us - https://aquigenbio.com/contact-us/
"The discovery of nitroso impurities in Crizotinib is concerning, as it highlights the critical need for rigorous quality control in drug manufacturing processes. We are dedicated to advancing our testing methodologies to detect and mitigate such impurities effectively. Our team is collaborating closely with regulatory authorities and pharmaceutical partners to address these issues transparently and swiftly. The health and safety of patients must remain our top priority, and we will continue to advocate for stringent measures that ensure the integrity of life-saving medications like Crizotinib. We are determined to play a leading role in enhancing drug safety for the benefit of all patients who rely on these essential therapies." - CEO, Aquigen Bio Sciences (Patient Safety and Communication)
For patients currently receiving Crizotinib treatment, understanding the potential risks of nitroso impurities is crucial. Healthcare providers are encouraged to communicate openly with their patients about the presence of these impurities and what it means for their treatment. Clear communication helps patients make informed decisions about their care, including whether to continue their current treatment or explore alternative options.
While the presence of nitroso impurities in Crizotinib is concerning, it is important to emphasize that the drug remains a critical treatment for many cancer patients. Treatment decisions should be made in consultation with healthcare providers, who can assess the risks and benefits of continuing therapy.
The Bottom Line:
The discovery of crizotinib nitroso impurities marks a pivotal moment for the pharmaceutical industry and cancer treatment. As regulators and pharmaceutical companies work to address this issue, patient safety remains the top priority. Healthcare providers must stay informed about the latest developments and communicate the risks and options to their patients. With collaborative efforts between industry experts, regulators, and medical professionals, the goal is to ensure that life-saving medications like Crizotinib remain safe, effective, and accessible for those who need them most.
Similar Trending Products:
1. N-Nitrosodiethylamine: https://aquigenbio.com/product/n-nitrosodiethylamine/
2. N-Nitrosodimethylamine: https://aquigenbio.com/product/n-nitrosodimethylamine-2/
3. N-Nitrosodimethylamine-d6: https://aquigenbio.com/product/n-nitrosodimethylamine-d6/
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a research organization based in Pune, India, specializing in a wide range of scientific research and development services. With a focus on innovation and excellence, Aquigen Bio Sciences delivers high-quality solutions to meet the complex needs of the pharmaceutical, environmental, and industrial sectors. The company is dedicated to advancing scientific knowledge and improving safety through cutting-edge research and technology.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Crizotinib Nitroso Impurity: Understanding the Potential Risks, Safety Concerns, and Their Impact on Cancer Treatment Protocols, Patient Outcomes, and Long-Term Safety Standards in Oncology Care here
News-ID: 3685216 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
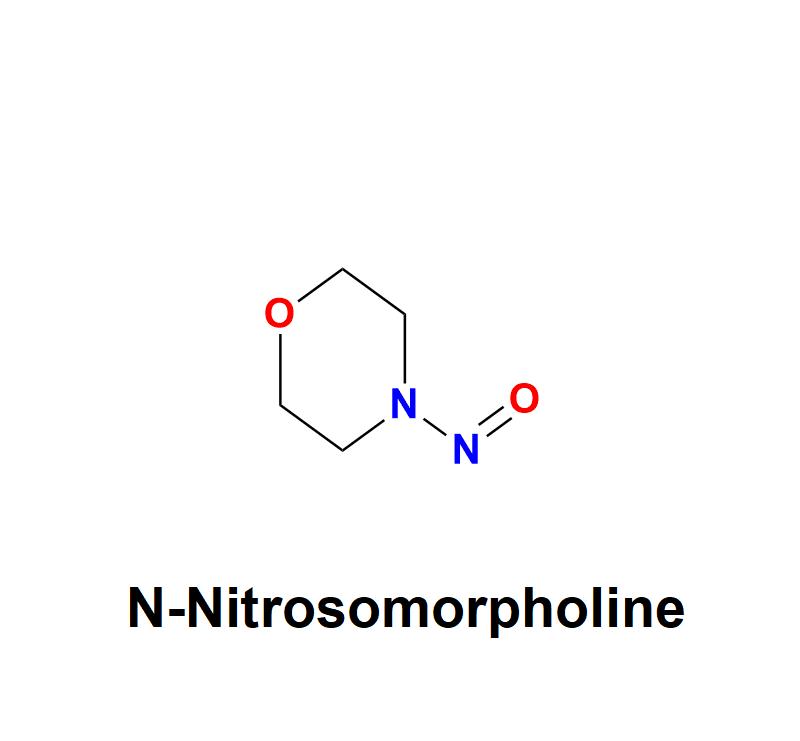
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
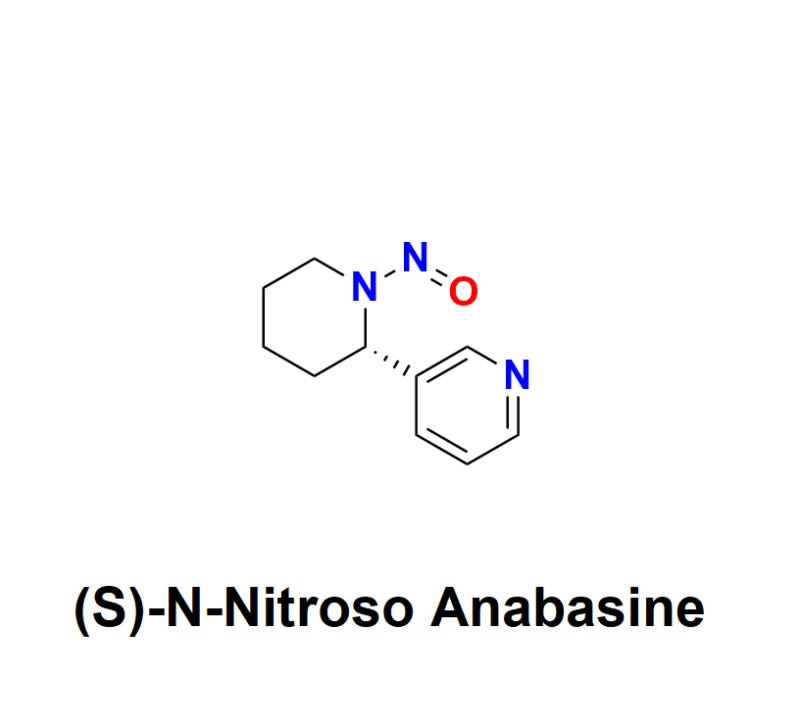
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
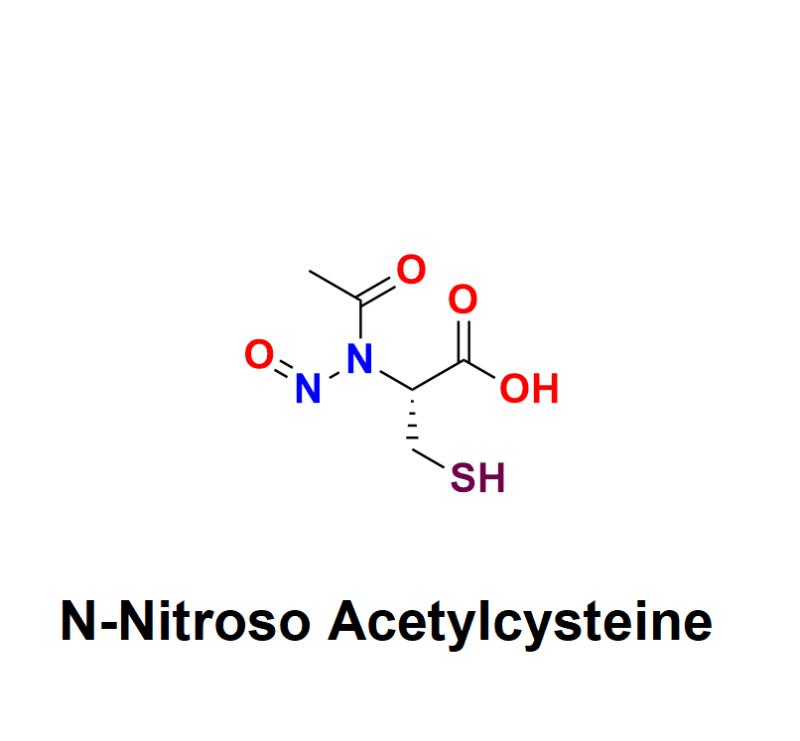
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…
More Releases for Crizotinib
Europe Crizotinib Market Size, Growth Factors, Historical Analysis and Industry …
Introduction:
The global Crizotinib market is experiencing significant growth, driven primarily by the increasing prevalence of non-small cell lung cancer (NSCLC) harboring specific genetic alterations. Crizotinib, a tyrosine kinase inhibitor, has emerged as a crucial therapy for patients with ALK-positive and ROS1-positive NSCLC. Several factors contribute to the market's expansion, including the rising incidence of these specific NSCLC subtypes, advancements in diagnostic techniques that allow for more accurate and timely identification…
Crizotinib Market Size is growing at a CAGR of 5.6% Forecast from 2025 - 2031 an …
Introduction:
The Crizotinib market is experiencing substantial growth, driven by its critical role in targeted cancer therapies, particularly for non-small cell lung cancer (NSCLC). This growth is underpinned by several key factors including increasing prevalence of genetic mutations such as Anaplastic Lymphoma Kinase (ALK) and ROS1, advancements in diagnostic technologies allowing for more precise patient selection, and the continued need for effective therapeutic interventions. Technological advancements in drug delivery mechanisms, along…
Crizotinib Capsules Market Outlook and Future Projections for 2030
The crizotinib capsules market represents a dynamic and continually evolving landscape, shaped by changing consumer demands and technological advancements. In this comprehensive report, we provide an in-depth exploration of the market, designed for a wide range of stakeholders including manufacturers, suppliers, distributors, and investors. Our goal is to equip industry participants with essential insights that enable informed decision-making in an ever-changing market environment. This analysis not only examines the current…
Crizotinib Market Size ,Growth Research Report 2024-2026 | 250 Pages
This Crizotinib market research provides a comprehensive study of the industry's size, share, demand, growth, gross profits, earnings, and sales. It analyzes the data acquired to provide a comprehensive perspective of industry trends and forecasts probable developments throughout the anticipated time period. For this study, historical data from 2016 and 2017 was collected, with 2018 serving as the base year to forecast the industry's growth from 2019 to 2026.
The global…
Crizotinib Market Scope, Future Prospects And Competitive Analysis 2021 to 2027 …
Collection analysis of data is depicted in this Crizotinib market research regarding efficient strategies, technological advances and market growth. Individual aspects and their interaction in the current market environment are understood by industry players easily with the help of relevant data provided in this market report. It further aims at covering statistical data to understand its operations. This Crizotinib market research covers needed modifications essential for businesses in developing and…
Crizotinib Market Report- Latest trending report is booming globally by Top key …
The Crizotinib Market research report highlights market insights with key trends and a breakdown of the XYZ market. In addition, Crizotinib Market provides information about products and offerings and impact due to macroeconomics and Covid-19 impact on the Crizotinib Market. Qualitative and quantitative insights are evaluated in the research report, and the Crizotinib Market size, growth, and share are also included in the research report. It also offers analysis…