Press release
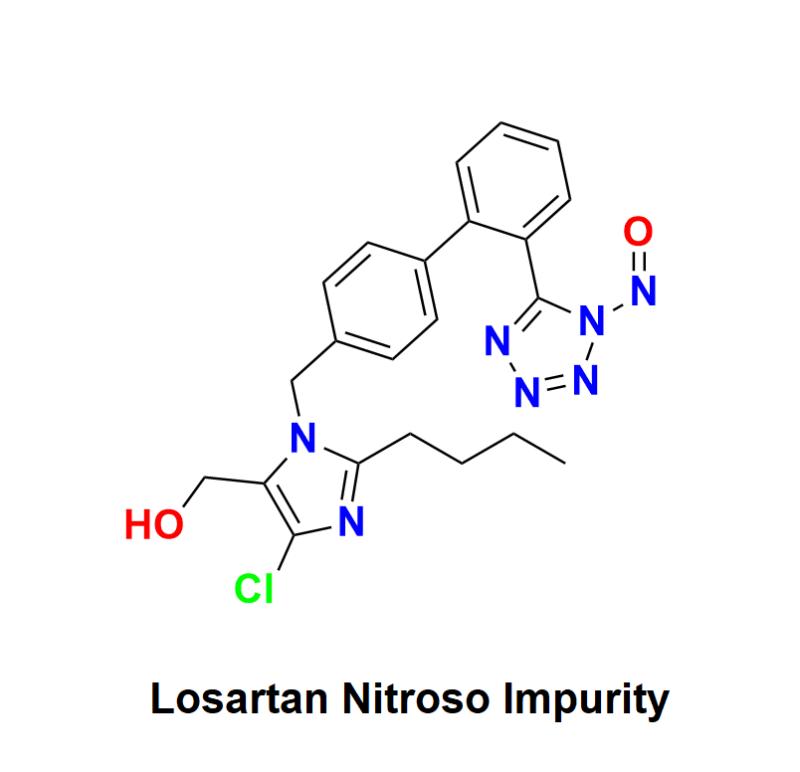
Losartan Nitroso Impurity: Addressing Compliance Risks, Regulatory Challenges, and Innovative Testing Solutions with Aquigen Bio Sciences to Ensure Patient Safety and Uphold Quality Standards in the Pharmaceutical Industry
Losartan Nitroso Impurity has emerged as a significant concern in the pharmaceutical industry as it faces mounting scrutiny regarding nitroso impurities. This issue has raised alarm among manufacturers and regulatory bodies alike. This press release aims to explore the challenges posed by Losartan Nitroso Impurity, outline the evolving regulatory landscape, and highlight the latest innovations in impurity testing that can help pharmaceutical companies mitigate compliance risks while ensuring patient safety.Understanding Losartan and Its Importance in Hypertension Management:
Losartan, an angiotensin II receptor blocker (ARB), is widely prescribed for managing hypertension and protecting kidney function in diabetic patients. Its effectiveness in lowering blood pressure and reducing cardiovascular risk makes it a critical medication for millions. However, the increasing awareness of nitroso impurities poses serious risks to both patient safety and the compliance landscape of pharmaceutical manufacturers.
Learn more about Losartan Nitroso Impurity: https://aquigenbio.com/product/losartan-nitroso-impurity/
The Challenge of Losartan Nitroso Impurities:
Nitroso impurities, classified as potential carcinogens, can inadvertently form during the synthesis, storage, or handling of pharmaceutical products. Detecting and quantifying these impurities presents unique challenges for manufacturers, as they are often difficult to identify at trace levels. The emergence of Losartan Nitroso Impurity underscores the need for a comprehensive approach to risk management within the pharmaceutical industry.
The presence of nitroso impurities in Losartan raises serious safety concerns for patients. With regulatory bodies intensifying their vigilance, pharmaceutical companies must navigate increased scrutiny and potential repercussions, including product recalls, regulatory actions, and damage to their reputations.
Regulatory Landscape: Navigating Compliance Challenges:
The regulatory environment concerning Losartan Nitroso Impurity has evolved significantly in recent years, driven by a growing recognition of the risks associated with nitroso contaminants. Regulatory agencies, including the FDA (U.S. Food and Drug Administration) and EMA (European Medicines Agency), have issued comprehensive guidelines aimed at assisting pharmaceutical manufacturers in identifying, assessing, and managing these impurities. These guidelines emphasize the necessity of establishing robust testing protocols specifically for Losartan, conducting thorough risk assessments, and ensuring meticulous documentation to maintain compliance with stringent safety standards.
Pharmaceutical companies must remain proactive in their compliance efforts to mitigate the risks associated with nitroso impurities. Failing to adequately address these concerns can result in serious consequences, including regulatory penalties, financial losses, and diminished public trust in the safety of their products.
Get in Touch with Us - https://aquigenbio.com/contact-us/
Innovative Practices for Mitigating Compliance Risks:
To navigate the complexities associated with Losartan Nitroso Impurity and other nitroso contaminants, pharmaceutical companies can implement a range of innovative practices aimed at ensuring compliance and upholding safety standards:
1. Advanced Analytical Testing: Utilizing state-of-the-art analytical techniques, such as liquid chromatography-tandem mass spectrometry (LC-MS/MS) and gas chromatography-mass spectrometry (GC-MS), enables manufacturers to detect and quantify nitroso impurities at trace levels, identifying potential risks early in the development process.
2. Comprehensive Risk Assessments: Conducting thorough evaluations of raw materials, manufacturing processes, and storage conditions is essential for proactively addressing risks and developing effective control strategies for nitroso impurities.
3. Cross-Department Collaboration: Encouraging collaboration between departments such as quality assurance, regulatory affairs, and research and development fosters a comprehensive compliance framework. Enhanced communication helps align practices with regulatory expectations.
4. Continuous Monitoring Protocols: Implementing continuous monitoring and testing throughout the manufacturing process allows for early detection of nitroso impurities, ensuring compliance with established safety standards before products reach the market.
5. Supplier Quality Management: Establishing stringent supplier quality management systems and conducting regular audits ensures the quality of raw materials and active pharmaceutical ingredients (APIs), minimizing the risk of contamination.
Upholding Safety Standards: Protecting Patients and Maintaining Trust
Patient safety remains paramount for pharmaceutical companies. The presence of nitroso impurities necessitates immediate and proactive measures to mitigate associated risks. By integrating innovative practices and fostering a culture of compliance, companies can demonstrate their commitment to patient safety while enhancing the overall quality of their products.
Conclusion:
As the pharmaceutical industry navigates the intricate landscape of nitroso contaminants, addressing compliance risks associated with Losartan Nitroso Impurity is crucial. By integrating innovative testing practices, conducting thorough risk assessments, and promoting interdepartmental collaboration, companies can uphold safety and regulatory standards while maintaining patient trust. A commitment to proactive compliance strategies will not only safeguard public health but also contribute to the long-term success and reputation of pharmaceutical organizations in a competitive market.
Similar Trending Products:
1. N-Nitroso Anatabine: https://aquigenbio.com/product/n-nitroso-anatabine/
2. N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/
3. N-Methyl N-Nitroso p-toluenesulfonamide: https://aquigenbio.com/product/n-methyl-n-nitroso-p-toluenesulfonamide/
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a premier contract research organization located in Pune, India, dedicated to providing cutting-edge solutions for impurity standards and regulatory compliance in the pharmaceutical industry. Specializing in the analysis and management of nitroso impurities, including Losartan Nitroso Impurity, Aquigen Bio Sciences is committed to enhancing safety and quality in drug development. By partnering with pharmaceutical companies, Aquigen Bio Sciences navigates the regulatory landscape, ensuring that patient care remains the top priority while addressing the complexities associated with nitroso contaminants.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Losartan Nitroso Impurity: Addressing Compliance Risks, Regulatory Challenges, and Innovative Testing Solutions with Aquigen Bio Sciences to Ensure Patient Safety and Uphold Quality Standards in the Pharmaceutical Industry here
News-ID: 3708987 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
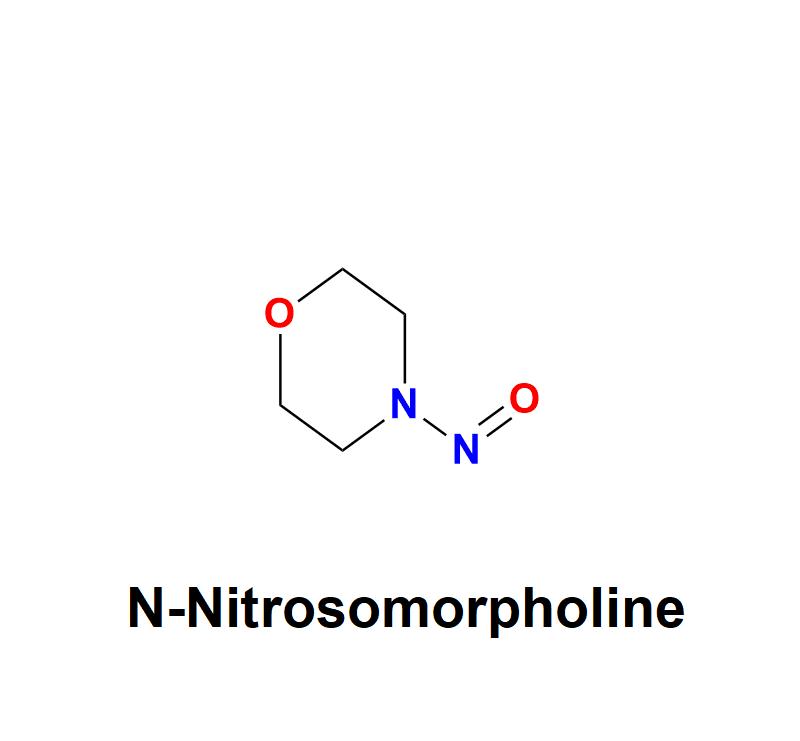
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
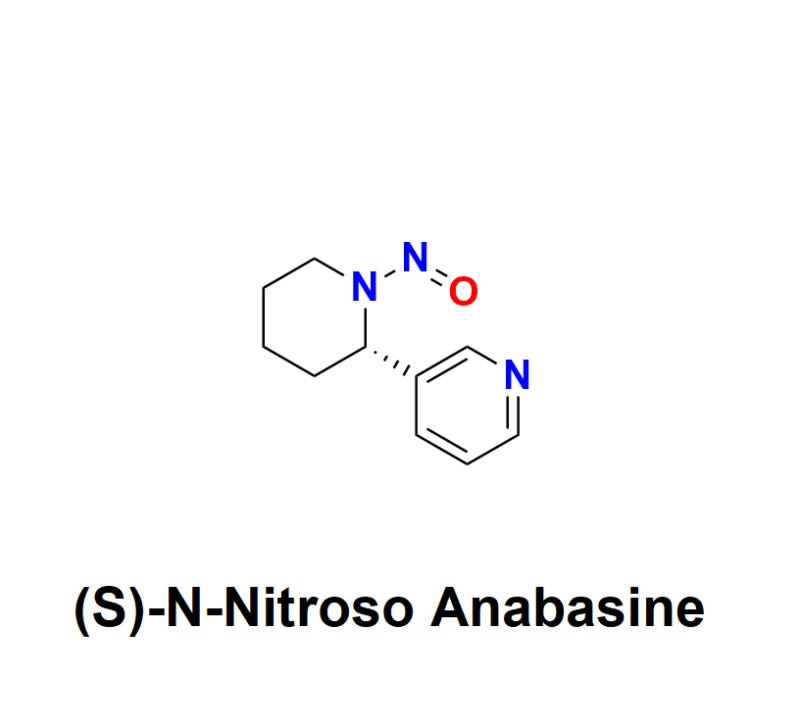
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
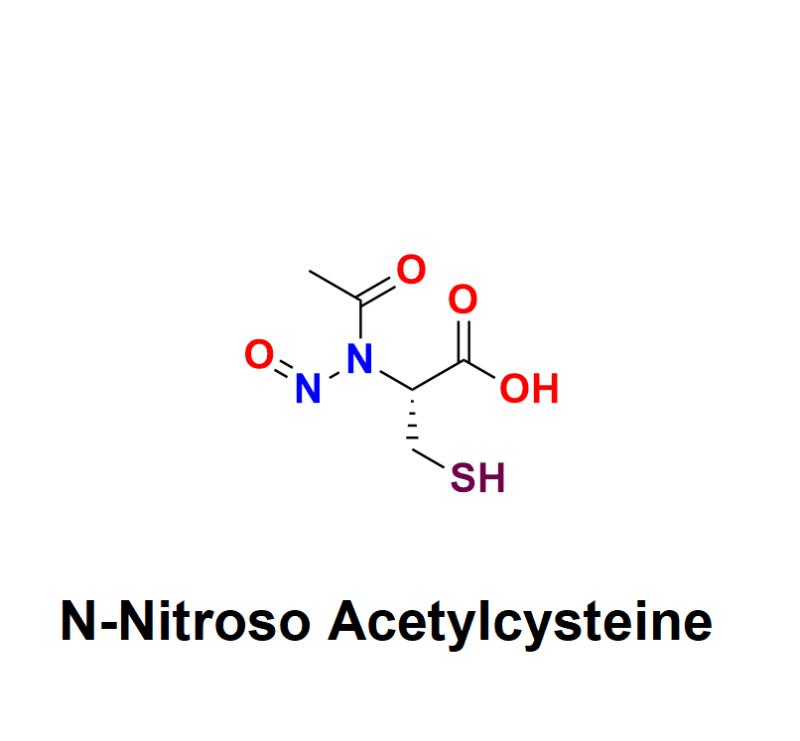
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…
More Releases for Losartan
Losartan Potassium Active Pharmaceutical Ingredient (API) Market Forecast 2025-2 …
Our market reports now include the latest updates on global tariffs, trade impacts, and evolving supply chain dynamics.
What Is the Projected Growth of the Losartan Potassium Active Pharmaceutical Ingredient (API) Market?
The market size of the losartan potassium active pharmaceutical ingredient (API) has seen impressive growth in the past few years. The prediction is that it will expand from $1.75 billion in 2024 to about $1.87 billion in 2025, showing a…
Losartan Industry Report 2025-2034: Market Dynamics, Trends, And Forecasts
Our market reports now include the latest updates on global tariffs, trade impacts, and evolving supply chain dynamics._x000D_
_x000D_
What Is the Expected Losartan Market Size During the Forecast Period?_x000D_
In previous years, the market size of losartan has been on a constant rise. From $1.44 billion in 2024, it's set to inflate to $1.50 billion in 2025, experiencing a compound annual growth rate (CAGR) of 4.6%. There are multiple factors leading to…
Losartan Potassium And Hydrochlorothiazide Tablet Market Size by Type, Applicati …
According to Market Research Intellect, the global Losartan Potassium And Hydrochlorothiazide Tablet market under the Internet, Communication and Technology category is expected to register notable growth from 2025 to 2032. Key drivers such as advancing technologies, changing consumer behavior, and evolving market dynamics are poised to shape the trajectory of this market throughout the forecast period.
The Losartan Potassium and Hydrochlorothiazide tablet market is witnessing consistent growth, primarily due to the…
Losartan Market Size by Type, Application, and Regional Outlook
USA, New Jersey- According to Market Research Intellect, the global Losartan market in the Internet, Communication and Technology category is projected to witness significant growth from 2025 to 2032. Market dynamics, technological advancements, and evolving consumer demand are expected to drive expansion during this period.
The Losartan market is experiencing steady growth, driven by the increasing prevalence of hypertension and cardiovascular diseases worldwide. As one of the most commonly prescribed angiotensin…
Losartan Potassium Hydrochlorothiazide Tablet Market Will Generate Record Revenu …
The Losartan Potassium Hydrochlorothiazide Tablet Market size is expected to grow at an annual average of CAGR 4% during the forecast period (2023-2029). Losartan potassium hydrochlorothiazide tablets are the first compound formulation of angiotensin II receptor (AT1) antagonists and diuretics. Suitable for patients with hypertension treated with combination medication.
Losartan Potassium Hydrochlorothiazide Tablet Market research report is the detailed investigation of the current market scenario and past issues to lessen…
Losartan Market Will Grow at a Healthy CAGR by 2023
The study report on the Losartan Market has assessed the historical and current performance of this market, especially highlighting the key trends and growth opportunities. It also gives the SWOT analysis and global revenue and sales 2018-2023.
According to the latest research, the rising demand for this product is driving the Losartan Market significantly. The expansion in the various related industry is also expected to reflect positively on the sales of…