Press release
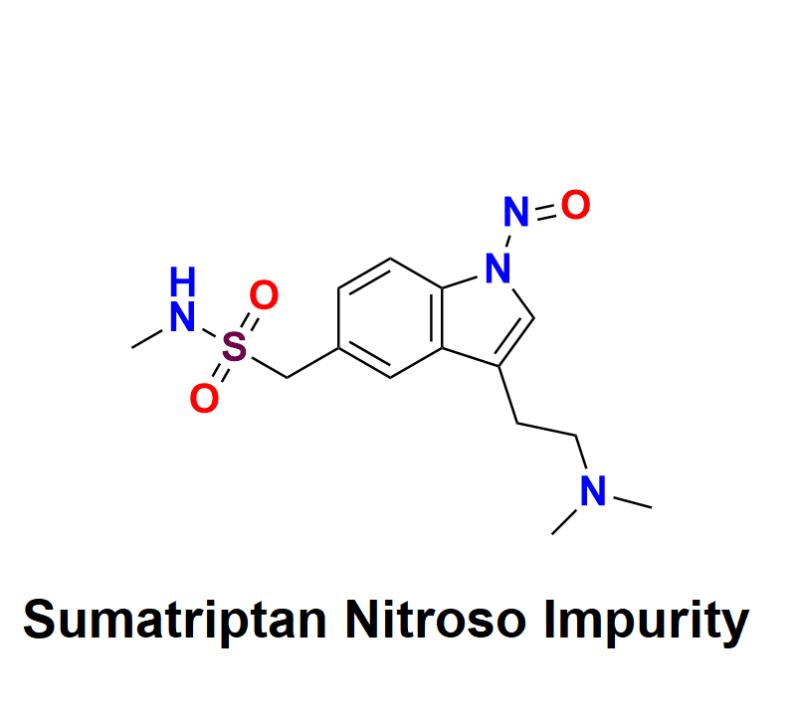
Sumatriptan Nitroso Impurity: Navigating Compliance Challenges and Implementing Innovative Solutions to Ensure Safety and Regulatory Adherence for Pharmaceutical Companies in the Evolving Landscape of Nitroso Contaminants in Migraine Medications
Sumatriptan Nitroso Impurity has become a pressing concern in the pharmaceutical industry, as companies grapple with significant compliance challenges related to these potentially harmful contaminants. As Sumatriptan, a widely prescribed medication for migraine relief, comes under increasing scrutiny from regulatory bodies, pharmaceutical companies must navigate a complex landscape to ensure product safety and adhere to evolving regulatory standards.What is the issue?
Nitroso impurities have emerged as a pressing concern within the pharmaceutical sector. Recent investigations have identified the presence of nitroso compounds in several medications, including Sumatriptan. These compounds can form during the manufacturing process, storage, or even from the drug's own degradation. Regulatory authorities, including the FDA and EMA, have set stringent limits on acceptable levels of these impurities, making it imperative for companies to address potential contamination proactively.
Learn more about Sumatriptan Nitroso Impurity: https://aquigenbio.com/product/sumatriptan-nitroso-impurity/
The rise in awareness regarding nitroso impurities stems from their association with increased cancer risk. This has led to heightened vigilance from regulatory agencies, which are now enforcing rigorous testing protocols. For pharmaceutical companies, this means revisiting their manufacturing processes and implementing robust testing frameworks to ensure compliance.
Compliance Challenges For Sumatriptan Nitroso Impurity:
1. Testing Protocols: Pharmaceutical companies are required to conduct thorough testing to detect nitroso impurities in their products. However, existing testing methods may not be adequately sensitive or specific enough to identify low-level contaminants. This raises questions about the reliability of current methodologies and highlights the need for advancements in analytical techniques.
2. Manufacturing Processes: The formation of nitroso impurities can occur at various stages of drug production, from raw material sourcing to final formulation. This complicates efforts to control contamination, as manufacturers must implement comprehensive risk assessments and control measures throughout the entire production process.
3. Regulatory Compliance: Compliance with evolving regulatory guidelines presents another layer of complexity. Pharmaceutical companies must stay informed about the latest regulatory expectations and ensure their processes align with these requirements. Failure to comply can result in costly recalls, legal implications, and damage to brand reputation.
4. Cost Implications: Implementing rigorous testing and control measures can significantly increase operational costs. Smaller companies, in particular, may struggle to allocate resources toward compliance efforts while maintaining competitive pricing for their products.
"As we confront the challenges posed by Sumatriptan Nitroso Impurity, I want to assure our partners that we are dedicated to providing innovative, comprehensive solutions. Together, we will ensure product safety, enhance regulatory compliance, and uphold the highest standards in the pharmaceutical industry for the benefit of public health," said the CEO of Aquigen Bio Sciences.
Get in Touch with Us - https://aquigenbio.com/contact-us/
Innovative Solutions for Sumatriptan Nitroso Impurity:
Despite these challenges, there are several innovative solutions that pharmaceutical companies can adopt to effectively address sumatriptan nitroso impurity concerns:
1. Enhanced Analytical Methods: Investing in advanced analytical techniques, such as ultra-high-performance liquid chromatography coupled with mass spectrometry (UHPLC-MS), can improve the sensitivity and specificity of nitroso impurity detection. These methods can facilitate the identification of trace levels of contaminants, enabling companies to meet regulatory standards more effectively.
2. Process Optimization: Companies should consider reviewing and optimizing their manufacturing processes to minimize the potential for sumatriptan nitroso impurity formation. This includes evaluating the choice of raw materials, reaction conditions, and storage conditions. By implementing stringent quality control measures, manufacturers can mitigate the risk of contamination.
3. Collaboration with Experts: Partnering with contract research organizations (CROs) and other industry experts can provide pharmaceutical companies with access to specialized knowledge and resources. Collaborations can help organizations develop effective testing protocols, conduct risk assessments, and ensure compliance with regulatory requirements.
4. Regulatory Intelligence: Staying informed about regulatory changes is crucial for compliance. Pharmaceutical companies should invest in regulatory intelligence platforms that provide real-time updates on guidelines, expectations, and best practices. This proactive approach can help companies adapt swiftly to new regulations.
5. Training and Education: Investing in employee training programs focused on compliance and quality assurance can significantly enhance a company's ability to manage sumatriptan nitroso impurity challenges. Educating staff about the risks associated with these impurities and the importance of adherence to protocols can foster a culture of compliance.
Conclusion:
As the pharmaceutical industry faces increasing scrutiny regarding nitroso impurities, particularly in widely used medications, companies must proactively address compliance challenges for sumatriptan nitroso impurity. By investing in enhanced analytical methods, optimizing manufacturing processes, collaborating with experts, staying informed about regulatory changes, and prioritizing training, pharmaceutical companies can navigate the complex landscape of nitroso impurities with confidence.
Similar Trending Products:
1. Brivanib Nitroso Impurity - https://aquigenbio.com/product/brivanib-nitroso-impurity/
2. Cilinidipine Nitroso Impurity - https://aquigenbio.com/product/cilinidipine-nitroso-impurity-2/
3. N-Nitroso Cilazapril - https://aquigenbio.com/product/n-nitroso-cilazapril/
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a research organization based in Pune, India, specializing in a wide range of scientific research and development services. With a focus on innovation and excellence, Aquigen Bio Sciences delivers high-quality solutions to meet the complex needs of the pharmaceutical, environmental, and industrial sectors. The company is dedicated to advancing scientific knowledge and improving safety through cutting-edge research and technology.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Sumatriptan Nitroso Impurity: Navigating Compliance Challenges and Implementing Innovative Solutions to Ensure Safety and Regulatory Adherence for Pharmaceutical Companies in the Evolving Landscape of Nitroso Contaminants in Migraine Medications here
News-ID: 3676332 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
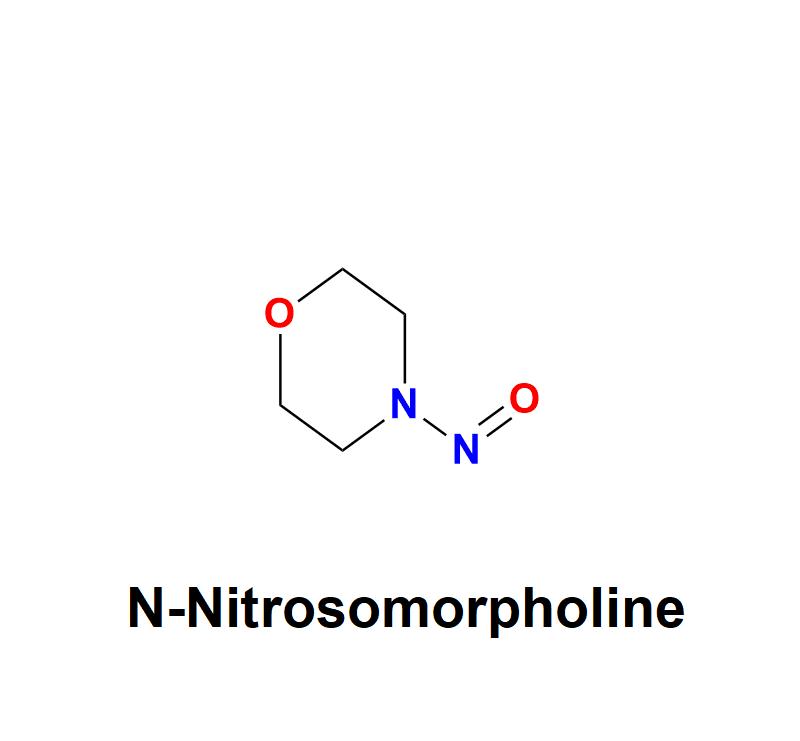
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
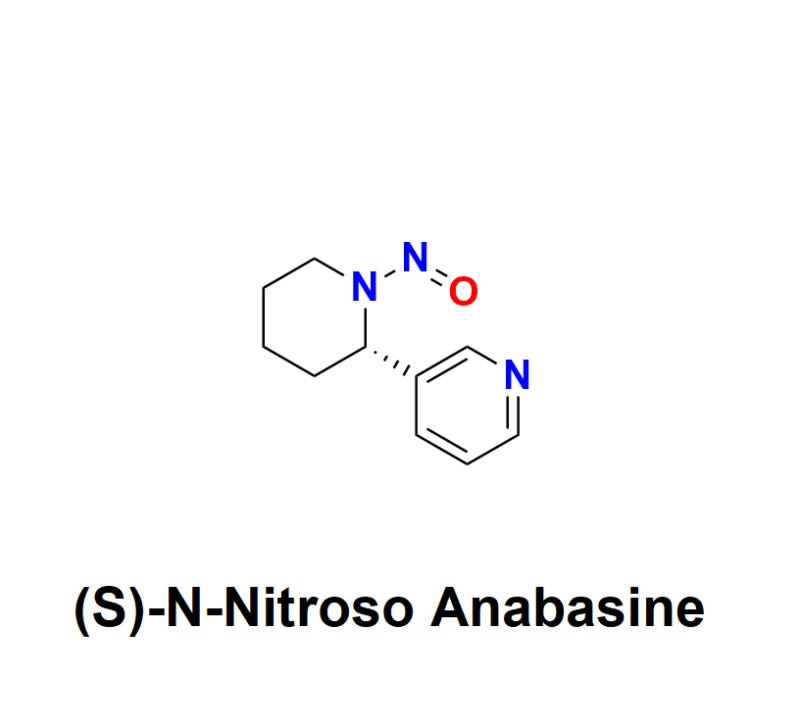
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
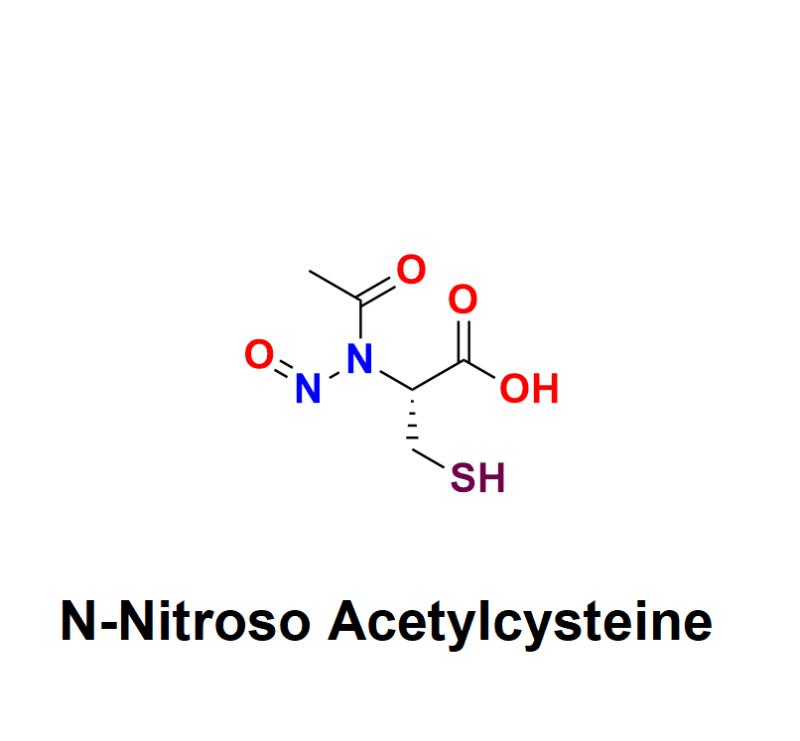
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…
More Releases for Nitroso
Riociguat N-Nitroso Des Formyl Impurity - Reliable Reference Standard for Advanc …
Riociguat N-Nitroso Des Formyl Impurity is a premium-quality analytical reference material developed to support research, testing, and quality control in the pharmaceutical industry.
As regulatory guidelines tighten worldwide, accurate detection, quantification, and control of nitroso impurities have become an essential part of drug development and manufacturing.
This compound plays a vital role in ensuring that formulations containing Riociguat meet the highest safety and compliance standards.
The Growing Importance of Nitroso Impurity…
Tizanidine Nitroso Impurity 1 - High-Purity Reference Standard for Reliable Impu …
In the highly regulated world of pharmaceutical manufacturing, impurity profiling plays a critical role in ensuring drug safety, efficacy, and regulatory compliance. Among the wide range of impurity reference standards required during API development and validation, Tizanidine Nitroso Impurity 1 stands out as a vital compound for researchers and manufacturers working on Tizanidine-based formulations.
At Aquigen Bio, we are proud to offer Tizanidine Nitroso Impurity 1, a high-purity reference standard meticulously…
Aquigen Bio Unveils N-Nitroso Felodipine: A New Benchmark in Pharmaceutical Refe …
In an era where drug safety and efficacy are paramount, the pharmaceutical industry faces increasing pressure to develop robust analytical methods that ensure the purity and quality of medicinal products. N-Nitroso Felodipine is specifically engineered to address these challenges. It serves as an indispensable tool for Analytical Method Development (AMD) and Analytical Method Validation (AMV), providing a reliable benchmark against which new and existing analytical procedures can be assessed for…
N-Nitroso Impurities: Carcinogenic Risks, Analytical Challenges, and Compliance …
N-Nitroso impurities are emerging as a significant concern in the pharmaceutical industry due to their potential carcinogenicity. These compounds, formed during the manufacturing or storage of drug products, have raised alarms among regulatory agencies across the globe. Pharmaceutical companies face mounting pressure to identify, analyze, and mitigate these impurities through stringent guidelines to ensure patient safety.
The serious risks posed by N-Nitroso compounds stem from their ability to induce genetic mutations…
Bumetanide Nitroso Impurity: Mitigating Compliance Risks and Integrating Innovat …
Bumetanide Nitroso Impurity has emerged as a significant concern in the pharmaceutical industry, particularly for companies involved in the development and manufacturing of medications used to manage edema. As awareness grows about the potential risks associated with nitroso impurities, regulatory agencies worldwide are increasing scrutiny over pharmaceutical products. This press release aims to address the challenges posed by Bumetanide Nitroso Impurity, outline the regulatory landscape, and highlight innovative practices that…
N-Nitroso-N-methylurea Market Expected to Develop by 2028 with COVID-19 Analysis
This N-Nitroso-N-methylurea market research report depicts evaluation methods, promotion methods, major updates, innovations and inventions. It enables business participants to track business profits, trends and latest market trends. It becomes easy for industry owners to set company goal through this market analysis report. The market statistics are obtained from secondary resources. Moreover, analysts perform detailed market study making use of a regression technique for attaining particular data about segment as…